Understanding the Critical Differences Between GLP-1 Weight Loss Drugs and Why Thousands Are Filing Lawsuits
Last Updated: January 2026
Introduction: Why Understanding These Differences Matters
If you’re researching GLP-1 weight-loss medications, you’ve likely encountered confusing brand names: Ozempic, Wegovy, Mounjaro, Zepbound, and others. You may be wondering:
- Is Ozempic the same as Wegovy?
- Why is Mounjaro called “next-generation”?
- Which medications are involved in lawsuits over severe side effects?
- Does it matter which one I took if I experienced complications?
These aren’t just medical questions; they have significant legal implications if you’ve suffered serious side effects.
While these drugs are often discussed together, they differ in crucial ways:
- Different active ingredients
- Different FDA-approved uses
- Different mechanisms inside your body
- Different manufacturers (and therefore different defendants in lawsuits)
This comprehensive guide will help you understand:
- The scientific differences between each medication
- Why all these drugs can cause similar severe complications
- How these differences affect your potential legal claim
- What steps to take if you’ve experienced serious side effects
Important Legal Note: If you developed gastroparesis, intestinal blockage, severe vomiting, or vision loss while taking any GLP-1 medication, you may have a valid legal claim regardless of which specific drug you used. Get a free case evaluation to understand your rights.
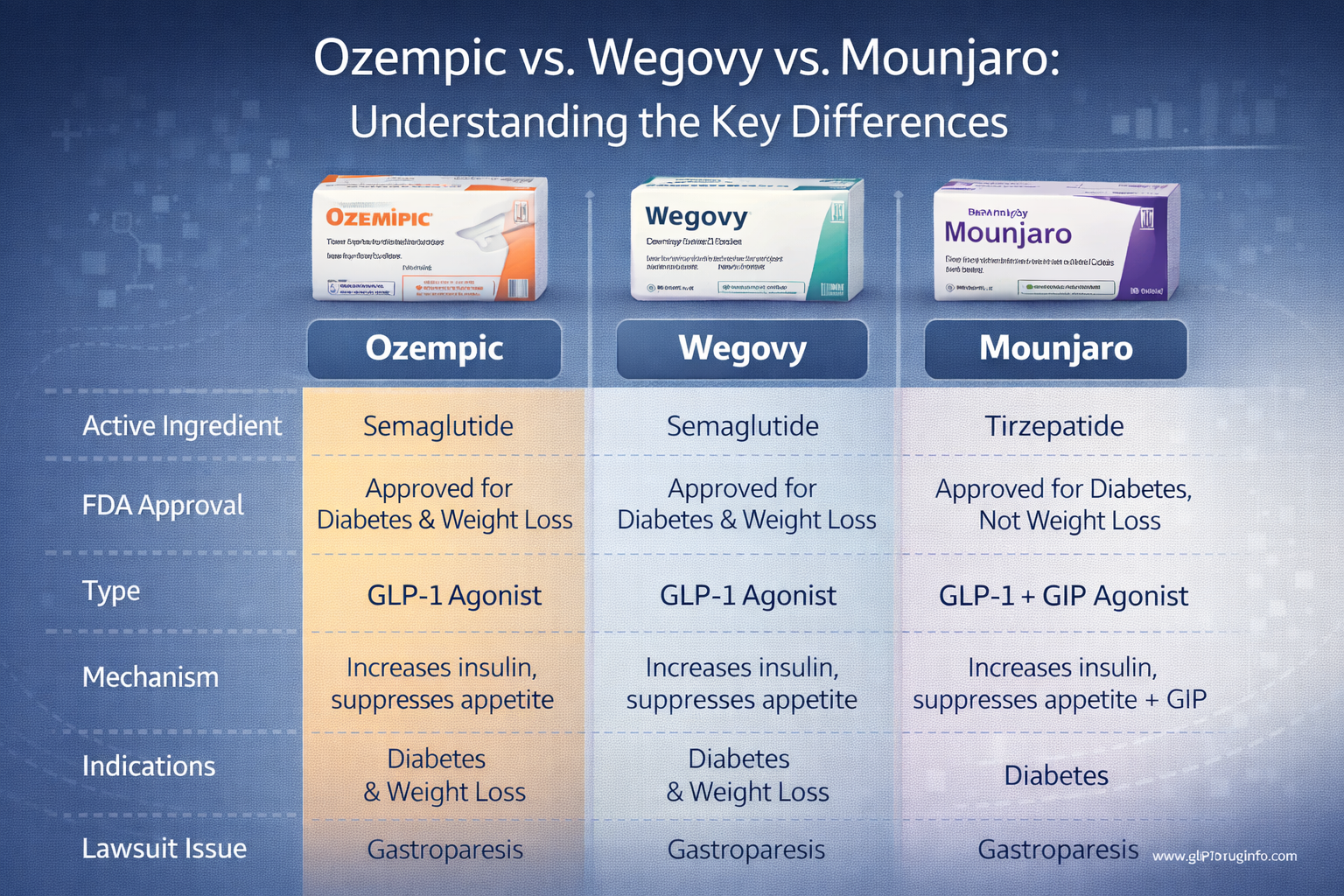
1. Active Ingredients: The Foundation of Each Drug
Every GLP-1 medication is defined by its active pharmaceutical ingredient - the compound that actually produces effects in your body. This determines:
- How the drug works
- What side effects you may experience
- Which manufacturer is responsible
- The specific legal arguments in your case
Semaglutide-Based Medications
Found in: Ozempic and Wegovy
Manufacturer: Novo Nordisk
How It Works:
- Mimics one natural hormone: GLP-1 (glucagon-like peptide-1)
- Triggers insulin release when blood sugar is elevated
- Slows gastric emptying (how fast food leaves your stomach)
- Suppresses appetite through brain receptors
- Long half-life allows once-weekly injection
Key Characteristics:
- Strong appetite suppression
- Proven track record (Ozempic approved 2017)
- Most extensively studied in clinical trials
- Highest number of adverse event reports
Clinical Effects:
- Average weight loss: 15-20% of body weight
- Significant reduction in A1C (blood sugar control)
- Cardiovascular benefits demonstrated in studies
Tirzepatide-Based Medications
Found in: Mounjaro and Zepbound
Manufacturer: Eli Lilly
How It Works:
- Mimics two natural hormones simultaneously:
- GLP-1 (glucagon-like peptide-1)
- GIP (glucose-dependent insulinotropic polypeptide)
- This “dual agonist” approach affects multiple metabolic pathways
- Also slows gastric emptying
- Enhances insulin secretion more powerfully
- Weekly injection schedule
Key Characteristics:
- Often produces greater weight loss than semaglutide
- Newer to market (Mounjaro approved 2022)
- Long-term safety data still evolving
- Complex dual-mechanism may affect risk profile
Clinical Effects:
- Average weight loss: 20-25% of body weight
- Stronger glucose control
- May work faster for some patients
Critical Warning: Because tirzepatide activates two hormone pathways instead of one, it may affect digestion in more complex ways. The mechanism that slows gastric emptying, believed to cause gastroparesis, is present in both semaglutide and tirzepatide drugs.
Why These Differences Matter for Your Legal Claim
The active ingredient determines:
-
Which manufacturer is the defendant
- Semaglutide cases: Novo Nordisk
- Tirzepatide cases: Eli Lilly
-
The timeline of what they knew and when
- Earlier drugs have longer history of adverse event reports
- Manufacturers may have had different levels of knowledge
-
The specific failure-to-warn arguments
- Different label warnings
- Different clinical trial data
- Different marketing claims
-
Causation analysis in your case
- How the drug caused your specific injury
- Whether dual-mechanism drugs pose different risks
For more detailed information: Learn about how GLP-1 drugs work in your body and what GLP-1 receptor agonists are.
2. Comprehensive Comparison: All GLP-1 Drugs at a Glance
Injectable GLP-1 Medications
| Brand Name | Active Ingredient | Type | Manufacturer | FDA Approved For | Approval Year | Max Dose | Lawsuit Status |
|---|---|---|---|---|---|---|---|
| Ozempic | Semaglutide | Single agonist (GLP-1) | Novo Nordisk | Type 2 Diabetes | 2017 | 2.0 mg weekly | In MDL 3094 |
| Wegovy | Semaglutide | Single agonist (GLP-1) | Novo Nordisk | Weight Management | 2021 | 2.4 mg weekly | In MDL 3094 |
| Mounjaro | Tirzepatide | Dual agonist (GLP-1/GIP) | Eli Lilly | Type 2 Diabetes | 2022 | 15 mg weekly | In MDL 3094 |
| Zepbound | Tirzepatide | Dual agonist (GLP-1/GIP) | Eli Lilly | Weight Management | 2023 | 15 mg weekly | In MDL 3094 |
| Victoza | Liraglutide | Single agonist (GLP-1) | Novo Nordisk | Type 2 Diabetes | 2010 | 1.8 mg daily | In MDL 3094 |
| Saxenda | Liraglutide | Single agonist (GLP-1) | Novo Nordisk | Weight Management | 2014 | 3.0 mg daily | In MDL 3094 |
| Trulicity | Dulaglutide | Single agonist (GLP-1) | Eli Lilly | Type 2 Diabetes | 2014 | 4.5 mg weekly | In MDL 3094 |
Oral GLP-1 Medications
| Brand Name | Active Ingredient | Manufacturer | FDA Approved For | Approval Year | Lawsuit Status |
|---|---|---|---|---|---|
| Rybelsus | Semaglutide (oral) | Novo Nordisk | Type 2 Diabetes | 2019 | In MDL 3094 |
Key Takeaways from This Comparison
1. Same Injury, Different Drugs All of these medications, despite different brand names, active ingredients, and manufacturers, share the critical mechanism of slowing gastric emptying. This is why all are involved in the same multidistrict litigation (MDL 3094) for gastroparesis and related injuries.
2. Diabetes vs. Weight Loss Labeling Notice that each active ingredient has two brand names:
- One for diabetes (Ozempic, Mounjaro, Trulicity)
- One for weight management (Wegovy, Zepbound, Saxenda)
This is purely a marketing distinction, the drugs work the same way. However, it matters legally because:
- Different labels have different warnings
- Off-label use (using Ozempic for weight loss) creates specific legal issues
- Your doctor’s prescription details affect your case
3. Newer Doesn’t Mean Safer Mounjaro and Zepbound are newer and may produce faster weight loss, but:
- They use the same gastric-slowing mechanism
- Long-term safety data is still limited
- Early reports suggest similar serious complications
- Being newer means manufacturers had access to earlier safety signals from semaglutide
4. Multiple Manufacturers = Multiple Defendants
- Novo Nordisk makes: Ozempic, Wegovy, Victoza, Saxenda, Rybelsus
- Eli Lilly makes: Mounjaro, Zepbound, Trulicity
If you took medications from both manufacturers, your legal case may involve both companies.
3. Ozempic vs. Wegovy: Same Drug, Different Purpose
The Most Common Question: Are They the Same?
The short answer: Yes, chemically identical.
The legal answer: No, treated as separate products with different risks and warnings.
What They Have in Common
Both Ozempic and Wegovy contain the exact same active ingredient: semaglutide. They are:
- Manufactured by the same company (Novo Nordisk)
- Administered the same way (weekly subcutaneous injection)
- Working through the same mechanism (GLP-1 receptor activation)
- Causing the same metabolic effects (insulin regulation, appetite suppression, slowed digestion)
The medication in a 2.0 mg Ozempic pen is molecularly identical to the medication in a Wegovy pen - there is no chemical difference whatsoever.
How They Differ
The differences are entirely in dosage, FDA approval, and marketing:
Ozempic (Diabetes Medication)
FDA-Approved Use:
- Treatment of Type 2 Diabetes Mellitus
- Reducing cardiovascular risk in diabetic patients
Dosing Schedule:
- Starting dose: 0.25 mg weekly (first 4 weeks)
- Maintenance dose: 0.5 mg or 1.0 mg weekly
- Maximum dose: 2.0 mg weekly
Intended Primary Effect:
- Blood sugar control
- A1C reduction
Weight Loss Status:
- Acknowledged as a “side effect”
- Not the primary indication
- Insurance may not cover for weight loss alone
Off-Label Use Reality: Many doctors prescribed Ozempic specifically for weight loss before Wegovy became available. This widespread off-label use creates specific legal questions about:
- Whether patients received adequate warnings
- Whether doctors were properly informed of risks
- Whether marketing encouraged inappropriate use
Wegovy (Weight Management Medication)
FDA-Approved Use:
- Chronic weight management in adults with obesity (BMI ≥30)
- Chronic weight management in adults with overweight (BMI ≥27) who have weight-related conditions
Dosing Schedule:
- Slower escalation schedule over 16-20 weeks
- Starting dose: 0.25 mg weekly
- Gradual increases: 0.5 mg → 1.0 mg → 1.7 mg
- Maximum dose: 2.4 mg weekly (higher than Ozempic)
Intended Primary Effect:
- Weight loss
- Body weight reduction of 15%+ on average
Additional Considerations:
- Typically requires obesity or overweight diagnosis
- May be covered by insurance for weight management
- Clinical trials specifically measured weight loss outcomes
Critical Legal Distinctions
Even though Ozempic and Wegovy are the same drug, in lawsuits your exact prescription matters significantly:
1. Label Warnings Differ
- Ozempic’s label emphasizes diabetes management and cardiovascular benefits
- Wegovy’s label emphasizes weight loss and obesity treatment
- The prominence and specificity of gastroparesis warnings may differ
- Failure-to-warn claims are based on the specific label language
2. Dosage Affects Risk Assessment
- Wegovy’s higher maximum dose (2.4 mg vs. 2.0 mg) may intensify side effects
- Some patients experienced gastroparesis at lower doses
- Others didn’t develop problems until higher doses
- Your specific dosage history is crucial evidence
3. Intended Use Matters
- Were you prescribed the drug for its FDA-approved purpose?
- Did your doctor prescribe Ozempic “off-label” for weight loss?
- Did you disclose your true purpose when obtaining the prescription?
- These factors may affect:
- What warnings should have been provided
- What you should have been told
- Whether your doctor shared liability
4. Different Marketing Claims
- Wegovy was aggressively marketed directly to consumers for weight loss
- Ozempic marketing technically focused on diabetes but implied weight benefits
- Social media influencers promoted both (often Ozempic off-label)
- Marketing materials affect what representations were made to you
Example Case Scenarios
Scenario A: Type 2 Diabetic on Ozempic
- Patient: 58-year-old with 10-year diabetes history
- Prescription: Ozempic 1.0 mg weekly for A1C control
- Result: Developed severe gastroparesis requiring feeding tube
- Legal claim: Even for approved use, warnings about gastroparesis were inadequate
Scenario B: Weight Loss Patient on Ozempic (Off-Label)
- Patient: 42-year-old seeking to lose 40 pounds, no diabetes
- Prescription: Ozempic 2.0 mg (prescribed off-label by weight loss clinic)
- Result: Chronic vomiting, diagnosed with gastroparesis
- Legal claim: Off-label promotion and inadequate warnings for weight-loss use
Scenario C: Obesity Patient on Wegovy
- Patient: 35-year-old with BMI 38, prescribed for weight management
- Prescription: Wegovy escalated to 2.4 mg
- Result: Intestinal blockage requiring emergency surgery
- Legal claim: Despite approved use, risks were not adequately disclosed
All three scenarios may have valid legal claims, but the specific arguments and evidence will differ.
What You Need to Document
If you took either Ozempic or Wegovy and suffered serious complications, gather:
-
Prescription records showing:
- Which exact brand name was prescribed (Ozempic or Wegovy)
- All dosages and dates
- Prescribing doctor and indication (diabetes vs. weight loss)
-
Medical records documenting:
- Your diagnosis before starting the medication
- Why the medication was prescribed
- What you were told about potential risks
- When symptoms began
- How your injury was diagnosed
-
Communication records:
- Doctor’s notes about discussing the medication with you
- Any written warnings or information sheets you received
- Pharmacy information provided with the prescription
For more details on gathering evidence: See our guide on How to Build a Strong Medical Injury Case: A Guide to Documenting GLP-1 Side Effects.
4. Mounjaro and Zepbound: The Dual-Agonist Difference
Why Tirzepatide Is Called “Next-Generation”
Mounjaro and Zepbound represent a fundamentally different approach to metabolic therapy. While semaglutide drugs mimic one hormone, tirzepatide mimics two hormones simultaneously.
The Dual-Agonist Mechanism Explained
Traditional GLP-1 Drugs (Single Agonist):
- Activate only GLP-1 receptors
- Trigger one metabolic pathway
Tirzepatide (Dual Agonist):
- Activates GLP-1 receptors (like semaglutide)
- ALSO activates GIP receptors (glucose-dependent insulinotropic polypeptide)
- Triggers two complementary metabolic pathways simultaneously
What Is GIP and Why Does It Matter?
GIP (Glucose-dependent Insulinotropic Polypeptide):
- Another incretin hormone produced in the intestines
- Stimulates insulin secretion from the pancreas
- Enhances fat metabolism
- Affects how the body stores and uses energy
- May amplify the appetite-suppressing effects of GLP-1
The Combined Effect: When both GLP-1 and GIP pathways are activated together:
- Stronger insulin response: better blood sugar control
- Enhanced fat burning: greater weight loss
- More appetite suppression: reduced food intake
- Slower gastric emptying: prolonged fullness (and potential problems)
Clinical Advantages of Tirzepatide
Greater Weight Loss: Clinical trials showed tirzepatide produced:
- Average weight loss: 20-25% of body weight
- Compared to semaglutide: 15-20%
- Some patients lost 30%+ of their starting weight
Faster Results: Many patients report:
- Quicker onset of appetite suppression
- More rapid weight loss in the first months
- Stronger “fullness” sensation after meals
Superior Glucose Control: For diabetic patients:
- Greater A1C reductions than semaglutide
- More patients reaching target A1C goals
- Fewer patients needing additional diabetes medications
Potential Disadvantages and Risks
1. Limited Long-Term Data
- Mounjaro only approved in 2022
- Zepbound only approved in 2023
- Long-term safety profile still emerging
- Unknown effects of decades-long use
2. More Complex Mechanism = More Complex Risks
- Activating two pathways may have unforeseen interactions
- GIP’s role in gastrointestinal function not fully understood
- Potential for different or additional side effects beyond GLP-1 alone
3. Severe Digestive Complications Despite being newer, tirzepatide drugs are already associated with:
- Gastroparesis (stomach paralysis)
- Ileus (intestinal blockage)
- Severe persistent vomiting
- Gastric outlet obstruction
- Cyclic vomiting syndrome
The mechanism is similar to semaglutide: excessive slowing of gastric emptying. However, the dual action may:
- Intensify this effect
- Make it more likely to occur
- Make it harder for the body to compensate
4. Higher Intensity Side Effects Even “common” side effects may be more severe:
- More intense nausea
- More frequent vomiting
- Greater gastrointestinal distress
- Harder adjustment period
Legal Implications: What Eli Lilly Knew
When Eli Lilly developed Mounjaro and Zepbound, they had a crucial advantage: knowledge from semaglutide’s track record.
By 2022, when Mounjaro was approved:
- Ozempic had been on the market for 5 years
- Reports of gastroparesis were accumulating
- Patient complaints were documented
- Medical literature was discussing concerns
This creates specific legal arguments:
1. Foreseeability Eli Lilly should have anticipated gastroparesis risk because:
- The same mechanism (slowed gastric emptying) was known from GLP-1 drugs
- Tirzepatide’s dual action might amplify this effect
- Clinical trial data should have been scrutinized for GI complications
2. Duty to Warn Despite this knowledge, plaintiffs allege:
- Warnings about gastroparesis were inadequate
- The permanence of stomach paralysis was understated
- Patients were not informed it could be irreversible
- Marketing emphasized weight loss benefits without balancing risks
3. Rushed to Market Some legal theories suggest:
- Pressure to compete with Wegovy’s success
- Inadequate safety monitoring in clinical trials
- Downplaying concerning signals to speed FDA approval
Mounjaro vs. Zepbound: The Same Story as Ozempic vs. Wegovy
Just like with semaglutide, tirzepatide has two brand names:
Mounjaro:
- FDA-approved for Type 2 Diabetes
- Maximum dose: 15 mg weekly
- Prescribed primarily for blood sugar control
Zepbound:
- FDA-approved for weight management
- Same maximum dose: 15 mg weekly
- Marketed for obesity treatment
The drugs are chemically identical, but the legal considerations mirror the Ozempic/Wegovy distinction:
- Different labels and warnings
- Different approved uses
- Potential off-label prescribing
- Different marketing claims
Case Example: Tirzepatide Injury
Patient Profile:
- 52-year-old woman with BMI 34
- No diabetes, seeking weight loss
- Prescribed Zepbound, escalated to 15 mg
Timeline:
- Month 1-3: Excellent weight loss (25 lbs), manageable nausea
- Month 4: Persistent vomiting began
- Month 5: Unable to eat solid food, lost 15 more lbs
- Month 6: Diagnosed with severe gastroparesis via gastric emptying scan
- Current: 12 months post-discontinuation, still cannot eat normally, requires liquid diet
Legal Claim:
- Inadequate warnings about permanence of gastroparesis
- Marketing emphasized dramatic weight loss without proper risk disclosure
- Failure to inform that condition might not resolve after stopping
- Damages include ongoing medical care, lost wages, diminished quality of life
5. Why All GLP-1 Drugs Face Lawsuits
Despite their differences in active ingredients, manufacturers, and mechanisms, all GLP-1 medications share one critical characteristic that makes them dangerous:
The Common Mechanism: Delayed Gastric Emptying
Normal Digestion:
- You eat food
- Your stomach churns and breaks it down (1-2 hours)
- Partially digested food moves into the small intestine
- Nutrients are absorbed
- Waste continues to the colon
On GLP-1 Medications:
- You eat food
- The drug dramatically slows stomach muscle contractions
- Food sits in the stomach for many hours (or days)
- Delayed movement into small intestine
- Prolonged fullness leads to appetite suppression and weight loss
For Most Patients: This causes temporary fullness and reduced appetite - the intended effect.
However, For Some Patients: This causes permanent damage to the stomach’s ability to empty:
- Gastroparesis (stomach paralysis)
- Food retention for 12-24+ hours
- Chronic vomiting
- Inability to digest solid food
- Malnutrition and dehydration
- Need for feeding tubes or IV nutrition
The Severe Complications Driving Lawsuits
1. Gastroparesis (Stomach Paralysis)
What It Is: A condition where the stomach’s muscles lose their ability to propel food forward normally, even after stopping the medication.
Symptoms:
- Severe nausea and vomiting
- Feeling full after eating very little (early satiety)
- Bloating and upper abdominal pain
- Heartburn and acid reflux
- Unpredictable blood sugar levels (in diabetics)
- Malnutrition and weight loss
Diagnosis: Requires objective testing, specifically a gastric emptying scan:
- Patient eats a standardized meal with radioactive tracer
- Imaging at 0, 1, 2, and 4 hours (the 4-hour measurement is crucial)
- If >10% of food remains at 4 hours, then gastroparesis diagnosis
Critical Legal Requirement: As of August 2025, Judge Marston’s ruling in MDL 3094 requires objective diagnostic testing (gastric emptying scan) to pursue a gastroparesis claim. Clinical diagnosis based only on symptoms is no longer sufficient. Learn more about gastric emptying scan requirements.
Why It’s Devastating:
- May be permanent even after stopping the drug
- Currently no cure, only symptom management
- Severely impacts quality of life
- May require years of treatment or permanent dietary changes
- Some patients need feeding tubes for life
Legal Significance: This is the primary injury in most GLP-1 lawsuits. Plaintiffs claim manufacturers:
- Knew or should have known gastroparesis could be permanent
- Failed to adequately warn patients and doctors
- Understated the severity and duration of risk
- Continued aggressive marketing despite safety signals
2. Ileus and Bowel Obstruction
What It Is: A partial or complete blockage of the intestines, or a temporary paralysis of intestinal movement.
How GLP-1 Drugs Cause It:
- Slowing effect extends beyond the stomach
- Affects small intestine and colon
- Food or stool becomes stuck
- Can be life-threatening without treatment
Symptoms:
- Severe abdominal pain and cramping
- Inability to pass gas or stool
- Severe bloating and distention
- Vomiting (may include fecal material)
- Loss of appetite
Treatment:
- Often requires emergency hospitalization
- Nasogastric tube to decompress the stomach
- IV fluids and electrolytes
- Some cases require emergency surgery
- Risk of intestinal perforation or death if untreated
Legal Claims: Patients who experienced bowel obstruction or ileus argue:
- This serious complication was not adequately disclosed
- Emergency room visits and surgeries could have been prevented
- Permanent bowel damage occurred
- Lost wages and medical bills are substantial
Learn more about ileus warning signs and symptoms.
3. Cyclic Vomiting Syndrome
What It Is: Recurring episodes of severe vomiting with no apparent cause, potentially triggered or worsened by GLP-1 medications.
Pattern:
- Sudden onset of intense nausea
- Vomiting that lasts hours to days
- Unable to keep down food or fluids
- Episodes may repeat weekly or monthly
- Between episodes, patient may feel normal
Impact:
- Frequent ER visits for IV rehydration
- Inability to work during episodes
- Dental damage from stomach acid
- Esophageal damage
- Severe disruption to daily life
Learn about about Vomiting Undigested Food: Identifying GLP-1–Induced Motility Disorders.
4. Severe Malnutrition and Dehydration
When GI Problems Become Systemic: Chronic inability to eat or keep food down leads to:
- Dangerous weight loss (beyond intended amount)
- Vitamin and mineral deficiencies
- Muscle wasting
- Weakness and fatigue
- Electrolyte imbalances (can cause heart problems)
- Need for IV nutrition (TPN - total parenteral nutrition)
Legal Damages:
- Cost of nutritional support
- Hospitalizations
- Lost ability to work
- Permanent health consequences
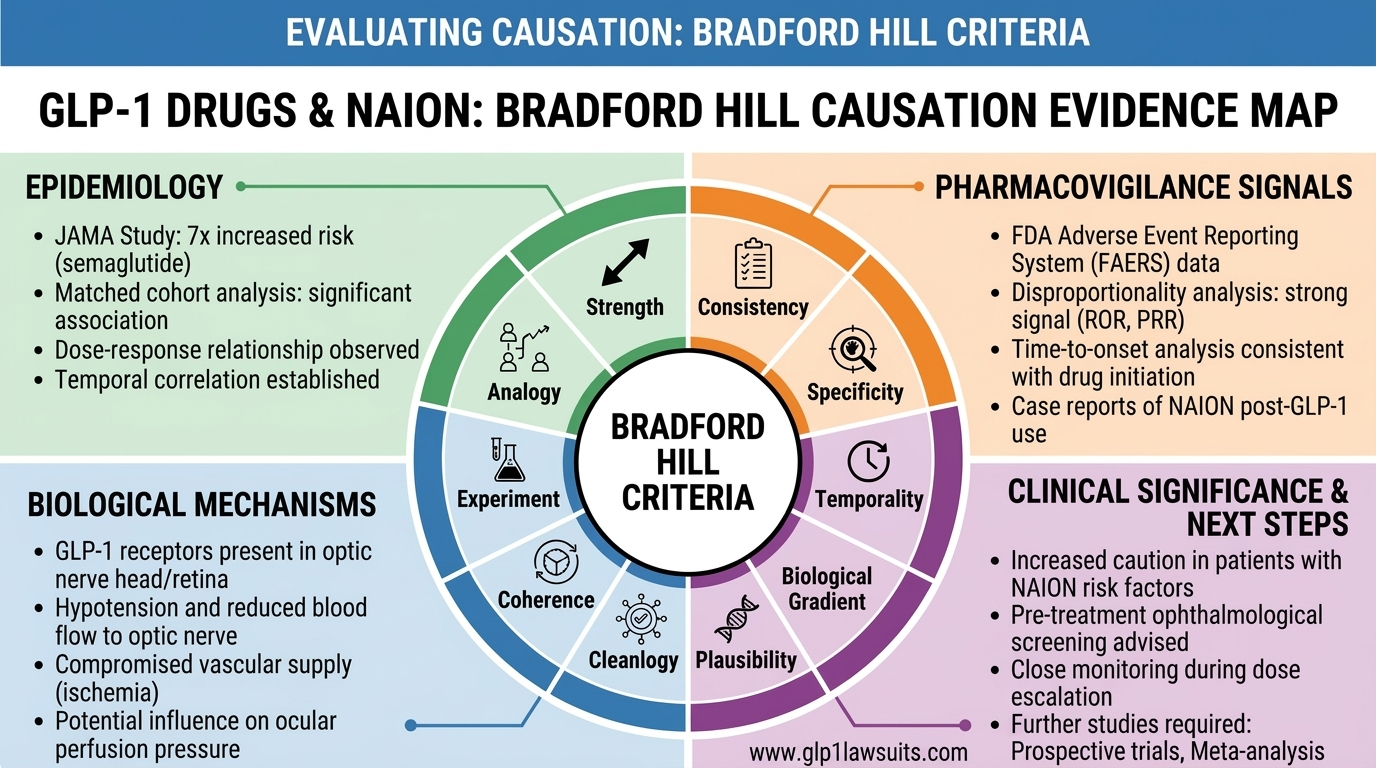
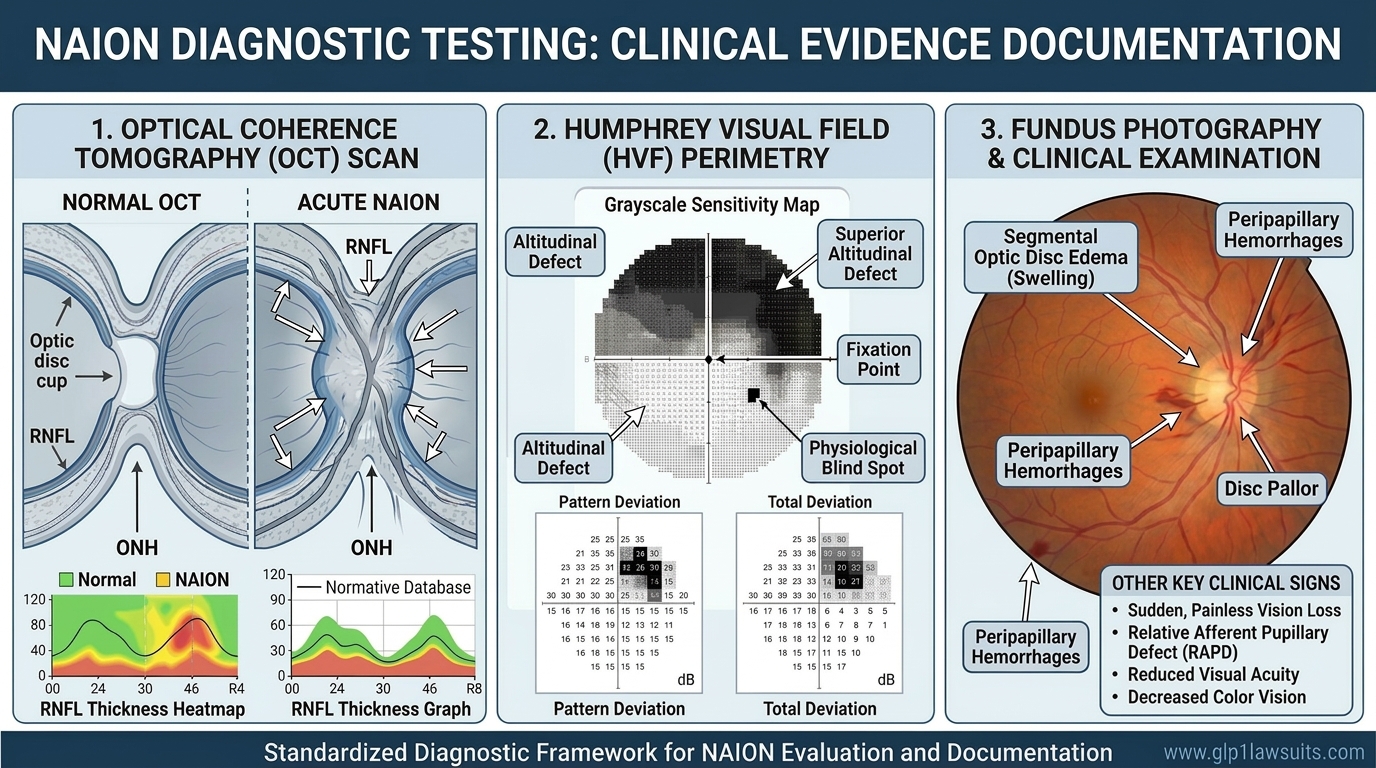
5. Sudden Vision Loss (NAION)
A Newly Recognized Serious Risk: Non-Arteritic Anterior Ischemic Optic Neuropathy (NAION), a form of eye stroke causing sudden vision loss.
What Patients Experience:
- Sudden vision loss in one or both eyes
- Usually painless
- Permanent in most cases
- May worsen over days to weeks
The Connection: Recent studies suggest GLP-1 drugs may increase NAION risk by 2-7 times. The mechanism is not fully understood, but theories include:
- Changes in blood flow to the optic nerve
- Rapid weight loss affecting ocular circulation
- Metabolic changes affecting eye health
Legal Development: Vision loss cases are being consolidated into a separate MDL (MDL 3163) also overseen by Judge Marston. This recognizes NAION as a distinct category of GLP-1 injury.
Detailed information on GLP-1 NAION and vision loss risks.
What All These Injuries Have in Common
Shared Characteristics:
- Underreported in clinical trials – The true incidence emerged only after millions of prescriptions
- Downplayed by manufacturers – Described as rare or temporary when they may be common and permanent
- Life-altering severity – These aren’t minor side effects; they permanently change patients’ lives
- Preventable with proper warnings – Many patients say they never would have taken the drug if properly informed
Shared Legal Theory: All lawsuits against Novo Nordisk and Eli Lilly center on failure to warn:
- Inadequate disclosure of severity
- Insufficient information about permanence
- Deceptive marketing that emphasized benefits over risks
- Failure to update warnings as adverse event data accumulated
The Litigation Landscape: MDL 3094
What Is an MDL? Multidistrict Litigation consolidates similar cases from across the country for pretrial proceedings. Learn more about what an MDL is.
MDL 3094 Status (January 2026):
- Presiding Judge: Hon. Karen S. Marston, Eastern District of Pennsylvania
- Number of Cases: Approximately 2,947 pending cases (and growing rapidly)
- Defendants: Novo Nordisk and Eli Lilly
- Covered Drugs: Ozempic, Wegovy, Mounjaro, Zepbound, Rybelsus, Victoza, Saxenda, Trulicity
Key Developments:
August 15, 2025 - Landmark Ruling: Judge Marston ruled that gastroparesis claims require objective diagnostic testing (gastric emptying scan). Clinical diagnosis based solely on symptoms is insufficient.
Impact: This ruling strengthened cases with proper medical documentation while potentially excluding some plaintiffs without formal testing.
Upcoming Timeline:
- March 27, 2026: Expert discovery deadline
- April 16, 2026: Summary judgment motions deadline
- Mid-2026: First bellwether trials expected to begin
Bellwether Trials: These are “test cases” selected to:
- Establish how juries react to the evidence
- Gauge potential verdict ranges
- Inform settlement negotiations
- Set precedents for remaining cases
Settlement Potential: While no global settlement has been reached, bellwether outcomes typically drive settlement discussions. Successful plaintiff verdicts could lead to settlement offers for the broader MDL.
6. Legal Differences: Does Your Specific Drug Matter?
While the core legal claims are similar across all GLP-1 medications, there are important distinctions based on which drug you took.
If You Took Semaglutide (Ozempic, Wegovy, Rybelsus, Victoza, Saxenda)
Defendant: Novo Nordisk
Advantages in Your Case: Longer market history = more extensive adverse event data
- Ozempic: On market since 2017
- Victoza: On market since 2010
- More documented cases of gastroparesis
- Stronger pattern of underreporting
-
Higher number of reported cases
- Semaglutide products have the most plaintiffs in MDL 3094
- More shared experiences and medical literature
- Strength in numbers for settlement negotiations
-
Clear timeline of manufacturer knowledge
- FDA adverse event reports available from 2017 onward
- Medical journal articles from 2019+
- Evidence of when Novo Nordisk knew or should have known
-
Off-label use concerns create additional claims
- Ozempic widely prescribed off-label for weight loss
- Marketing may have encouraged inappropriate use
- Inadequate warnings for weight-loss patients
Potential Challenges:
- Novo Nordisk will argue warnings were adequate for the time period
- They may claim patients were informed of GI risks
- Defense will emphasize that nausea/vomiting were disclosed
If You Took Tirzepatide (Mounjaro, Zepbound)
Defendant: Eli Lilly
Advantages in Your Case: Foreseeability argument
- By 2022, semaglutide’s gastroparesis risk was known
- Eli Lilly should have anticipated similar or worse effects
- Dual mechanism might amplify gastric slowing
-
Higher expectations for warning
- Later approval means higher standard
- Should have learned from Novo Nordisk’s experience
- Less excuse for inadequate warnings
-
Potentially stronger causation
- Dual agonist mechanism may show more direct link to injury
- Less time for alternative explanations
- Clearer temporal relationship if you developed symptoms quickly
-
Growing case numbers
- More patients reporting issues as time passes
- Pattern emerging of serious complications
- Eli Lilly facing increasing scrutiny
Potential Challenges:
- Newer drug means fewer total cases (so far)
- Less accumulated medical literature
- Defense may argue insufficient time to know about rare complications
- Dual mechanism creates more complex causation analysis
If You Took Multiple GLP-1 Drugs
This is common - many patients:
- Started on Ozempic, switched to Wegovy
- Tried Mounjaro after semaglutide didn’t work or caused side effects
- Were prescribed different drugs as insurance coverage changed
Legal Approach:
- Disclose all GLP-1 medications to your attorney
- Cases can still proceed with multiple drug exposure
- May pursue claims against both Novo Nordisk and Eli Lilly
- Causation analysis will examine cumulative exposure
Key Considerations:
- When did your symptoms begin relative to each drug?
- Did symptoms worsen when switching drugs?
- Was there a clear temporal relationship?
- Medical records must document timeline precisely
What Your Attorney Will Need to Know
Regardless of which medication(s) you took, be prepared to provide:
1. Prescription History:
- Exact brand name(s) prescribed
- All dosages and dose escalations
- Start and stop dates for each medication
- Prescribing physician(s)
- Medical indication (diabetes vs. weight loss vs. other)
- Any switches between drugs and why
2. Medical Records:
- Pre-treatment health status
- Diagnosis that led to prescription
- Documentation of symptoms
- Diagnostic testing (especially gastric emptying scans)
- Treatment for complications
- Hospitalizations and ER visits
- Current status and ongoing treatment
3. Informed Consent Documentation:
- What you were told about risks
- Any consent forms signed
- Information sheets provided by pharmacy
- Notes from doctor about discussion
- Whether gastroparesis was mentioned
4. Impact Documentation:
- Medical bills and expenses
- Lost wages and employment records
- Changes to diet and lifestyle
- Ongoing symptoms and limitations
- Effect on quality of life
Complete list of documents needed for your claim.
Bottom Line: All Drugs Are Covered in MDL 3094
The good news: Regardless of which specific GLP-1 medication you took, if you suffered serious injuries, you likely have a valid claim. The MDL structure allows for differences while pursuing the common theme of failure to warn.
The key requirement: You must have objective medical evidence of your injury, particularly for gastroparesis claims (gastric emptying scan showing delayed emptying at 4 hours).
7. Frequently Asked Questions
About the Drugs and Their Effects
Q: Is Wegovy more dangerous than Ozempic since the dose is higher?
A: Both medications contain the same active ingredient (semaglutide), so they carry the same types of risks. While Wegovy’s higher maximum dose (2.4 mg vs. 2.0 mg) may increase the intensity of side effects for some patients, serious injuries like gastroparesis have been reported at all dosage levels, ****including at the lowest doses.
The risk exists with both medications. Some patients developed severe gastroparesis at just 0.5 mg weekly, while others tolerated higher doses without major complications. Individual variation in gastric sensitivity appears to be a significant factor.
Key point: The dose difference between Ozempic and Wegovy is relatively small (2.0 vs. 2.4 mg). The legal claims focus not on which drug is “more dangerous,” but on whether warnings were adequate for both.
Q: Should I switch from Ozempic to Mounjaro to avoid side effects?
A: No. This would not reduce your risk and might increase it. Mounjaro (tirzepatide) works through a similar mechanism to Ozempic (semaglutide), both slow gastric emptying, which is the mechanism believed to cause gastroparesis. In fact, Mounjaro’s dual-agonist action (affecting both GLP-1 and GIP receptors) may potentially amplify digestive effects.
Switching between GLP-1 drugs does not eliminate the underlying mechanism causing these injuries. All drugs in this class carry similar risks of:
- Severe gastroparesis
- Intestinal blockage
- Persistent vomiting
- Other gastrointestinal complications
If you’re experiencing side effects:
- Report them to your doctor immediately
- Do not stop abruptly without medical guidance
- Consider whether continuing is worth the risks
- Discuss all alternatives including non-GLP-1 options for diabetes or weight management
- Get objective testing (gastric emptying scan) if you have severe GI symptoms
Better alternatives to discuss with your doctor:
- Traditional diabetes medications (metformin, insulin, SGLT2 inhibitors)
- Non-GLP-1 weight loss medications
- Lifestyle interventions with medical supervision
- Surgical options if appropriate
Q: Can I file a lawsuit if I took a GLP-1 drug for diabetes instead of weight loss?
A: Yes, absolutely. The lawsuits are not limited to weight-loss patients.
The legal claims focus on failure to adequately warn about severe side effects regardless of why the drug was prescribed. Both diabetes patients and weight-loss patients have valid claims if they developed serious injuries.
Why your prescription purpose doesn’t disqualify you:
- The injury is the same whether you took the drug for diabetes or weight loss
- The warnings were inadequate for both approved uses
- The mechanism is identical regardless of indication
- The failure to warn affected all patients, not just one category
In fact, diabetes patients may have stronger arguments:
- You needed the medication for a serious health condition
- You had fewer alternatives available
- The risk-benefit calculation was different
- You were a captive market with less choice
Your claim will focus on:
- Whether you were adequately informed of gastroparesis risk
- Whether the severity and potential permanence were disclosed
- Whether you would have chosen differently if properly warned
- The damages you suffered as a result
Q: What if I took multiple GLP-1 drugs over time?
A: You can still pursue a claim. Taking multiple GLP-1 medications is very common and does not disqualify you.
Common scenarios:
- Started on Ozempic for diabetes, later switched to Mounjaro for better glucose control
- Tried Victoza, then upgraded to Ozempic as newer options became available
- Used Ozempic off-label for weight loss, then switched to Wegovy when it was approved
- Prescribed Trulicity initially, changed to Mounjaro due to insurance coverage
What to disclose to your attorney:
-
Complete chronology of all GLP-1 drugs taken: - Names and dates for each medication - Dosages and how they changed over time - Reasons for switching
-
Timeline of symptoms:
- When did GI symptoms begin?
- Which drug(s) were you taking at that time?
- Did symptoms change when switching drugs?
- Did they worsen, improve, or stay the same?
-
Medical documentation:
- Prescription records for all medications
- Doctor’s notes about medication changes
- Records of reported side effects
- Diagnostic testing and when it was performed
How this affects your case:
If symptoms began with the first drug:
- Clear temporal relationship
- Strong causation argument
- May have claims against that manufacturer
If symptoms worsened with each subsequent drug:
- Cumulative damage theory
- May have claims against multiple manufacturers
- Important to show progression
If symptoms appeared only after starting the second or third drug:
- Still valid claim
- May need to address why earlier drug was tolerated
- Focus on the drug most directly linked to injury
Causation analysis: Your medical experts will examine:
- Which drug you were taking when symptoms began
- Whether cumulative exposure contributed
- Whether one drug’s effects sensitized you to another
- The biological plausibility of the timeline
Multiple defendants: If you took medications from both Novo Nordisk and Eli Lilly, you may have claims against both companies. Each manufacturer is responsible for adequately warning about their own product’s risks.
About Symptoms and Diagnosis
Q: Do I need a gastroparesis diagnosis to have a case?
A: Gastroparesis is the most common qualifying injury, but not the only one.
Other qualifying injuries include:
- Bowel obstruction or ileus (with imaging/medical records)
- NAION (sudden vision loss)
- Severe persistent vomiting requiring hospitalization
- Other serious GI complications requiring emergency intervention
However, for gastroparesis claims specifically:
Yes, you need formal diagnosis, and as of August 2025, objective testing is required.
Judge Marston’s August 15, 2025 ruling clarified:
- Clinical diagnosis based solely on symptoms is not sufficient
- You must have objective diagnostic testing
- Specifically: gastric emptying scan (4-hour scintigraphy study)
- Alternative tests: gastric emptying breath test or wireless motility capsule (SmartPill, though discontinued)
Why this matters:
- Many patients were told they had “gastroparesis” based only on symptoms
- Without objective testing, defendants will challenge the diagnosis
- Court ruled that symptom-based diagnosis isn’t reliable enough
- Need scientific proof of delayed gastric emptying
What if you have symptoms but no formal testing?
If you’re still experiencing symptoms:
- Get tested immediately - see a gastroenterologist
- Request a 4-hour gastric emptying scan
- Ensure results are properly documented
- Get formal written diagnosis
If your symptoms have resolved:
- More difficult to prove you had gastroparesis
- Current testing may show normal emptying
- May still have a case if you have:
- Medical records documenting severe symptoms during active use
- Hospitalization records
- Endoscopy reports showing food retention
- Doctor’s notes discussing gastroparesis
- But case will be weaker without objective diagnostic test
Bottom line for gastroparesis claims:
- Objective testing = required (gastric emptying scan)
- Clinical symptoms alone = not sufficient
- Test must show delayed emptying at 4-hour mark
- Get tested while symptomatic if possible
For other injuries (bowel obstruction, vision loss): Different objective evidence is required:
- Bowel obstruction: CT scan, X-ray, surgical records
- NAION: Ophthalmologist diagnosis, visual field testing, OCT imaging
Complete guide to gastric emptying scans and requirements.
Q: What if I’m still taking the medication but experiencing problems?
A: Your health comes first. If you’re experiencing serious side effects:
Immediate steps:
1. Contact your doctor immediately
- Report all symptoms honestly and completely
- Don’t minimize or downplay what you’re experiencing
- Ask whether continuing is safe
2. Do not stop abruptly without medical guidance
- Some medications require tapering
- Sudden discontinuation may cause other issues
- Your doctor needs to manage the transition safely
3. Get proper testing
- Request gastric emptying scan if you have severe GI symptoms
- Get imaging if you have abdominal pain
- See appropriate specialists (gastroenterologist, etc.)
- Document everything
4. Document your symptoms
- Keep a daily symptom diary
- Record what you can/cannot eat
- Note severity and frequency of vomiting
- Track weight changes
- Photograph any visible symptoms
Legal considerations while still on the medication:
Advantages of early action:
- Easier to prove causation – clear temporal relationship
- Testing while symptomatic – more likely to show abnormalities
- Fresh medical records – contemporaneous documentation
- Can show progression – symptoms while on drug vs. after stopping
Potential challenges:
- Continuing despite symptoms - defense may argue you assumed the risk
- Duty to mitigate - legally required to minimize damages by seeking appropriate medical care
What you should do legally:
Consult an attorney now:
- Even if you’re still on the medication
- Don’t wait until damage is irreversible
- Early legal advice can help you document properly
- Attorney can advise on medical decisions from legal perspective
Preserve evidence:
- Keep all prescription bottles and packaging
- Save pharmacy receipts and records
- Keep copies of all medical records
- Save any written materials from doctor or pharmacy
Financial considerations:
- Your medical expenses from complications may be recoverable
- Don’t delay treatment due to cost concerns
- Document all out-of-pocket costs
Making the decision to continue or stop:
Factors to discuss with your doctor:
- Severity of your current symptoms
- Whether symptoms are worsening or stable
- Alternative treatment options for your condition
- Risk of permanent damage if you continue
- Potential for symptom resolution if you stop
Your rights:
- You have the right to make informed medical decisions
- You can refuse to continue medication that’s harming you
- You can seek second opinions
- You can consult with specialists
Bottom line: Prioritize your health and safety. If you’re experiencing severe symptoms, work with your doctor to determine whether continuing is appropriate. Simultaneously consult a legal professional to understand your rights and ensure proper documentation.
Free case evaluation available here.
About Lawsuits and Legal Process
Q: How long do I have to file a lawsuit?
A: This depends on your state’s statute of limitations, but generally you have 2-4 years from when you discovered (or should have discovered) your injury.
When does the clock start?
Discovery Rule: Most states use the “discovery rule” - the statute starts when you:
- Knew or reasonably should have known you were injured
- Knew or should have known the injury was caused by the medication
- Had enough information to investigate a potential claim
Example timelines:
Scenario 1:
- Started Ozempic: January 2022
- Developed severe vomiting: March 2022
- Diagnosed with gastroparesis via gastric emptying scan: June 2022
- Statute likely starts: June 2022 (formal diagnosis)
- Deadline (in 2-year state): June 2024
Scenario 2:
- Started Wegovy: March 2023
- Had nausea but continued medication: 2023-2024
- Stopped medication: January 2025
- Symptoms persisted and worsened: Spring 2025
- Diagnosed with gastroparesis: July 2025
- Statute likely starts: July 2025 (when severity became clear)
- Deadline (in 2-year state): July 2027
Special considerations:
Continuing harm: Some courts apply “continuing tort” doctrine:
- If injury is ongoing and worsening
- Statute may not start until harm stops progressing
- This is state-specific and fact-dependent
Fraudulent concealment: If manufacturers actively hid risks:
- Statute may be “tolled” (paused)
- Could extend beyond normal limitations period
- Must prove active concealment, not just failure to warn
Minor children:
- Statute typically doesn’t start until child turns 18
- Then runs for normal period (2-4 years)
- Parents can sometimes file on behalf of minors sooner
Why you shouldn’t wait:
Evidence preservation:
- Medical records may be destroyed after certain periods
- Witnesses’ memories fade
- Physical evidence deteriorates
- Pharmacies may purge prescription records
MDL considerations:
- Cases filed early get into the MDL sooner
- Earlier filings may be considered for bellwether trials
- Settlement allocation may favor earlier filers
Strengthening your case:
- More time to gather evidence
- Ability to get proper diagnostic testing while symptomatic
- Documentation of progression over time
Risk of running out of time:
- Missing the statute of limitations = case permanently barred
- No exceptions or extensions in most cases
- Even meritorious claims are dismissed if filed too late
What to do right now:
Step 1: Find out your state’s statute Contact us and we’ll tell you the specific deadline for your state.
Step 2: Act immediately if you’re close to the deadline If your injury was diagnosed more than 18 months ago:
- You may be approaching your deadline
- Consult an attorney urgently
- Don’t delay even a few weeks
Step 3: Even if you have time, act now
- Earlier is always better
- Cases take time to prepare
- Don’t procrastinate
Bottom line: Statutes of limitations are absolute deadlines. When in doubt, consult an attorney immediately. A free consultation can clarify your timeline and ensure you don’t miss your window. The call costs nothing; missing your deadline costs everything.
Get Your Case Evaluated: Free Consultation
Whether you took Ozempic, Wegovy, Mounjaro, Zepbound, or another GLP-1 medication, if you developed serious complications, you may be entitled to significant compensation.
Qualifying Injuries Include:
Gastrointestinal Injuries:
- Gastroparesis (stomach paralysis) confirmed by gastric emptying scan
- Persistent vomiting requiring hospitalization or IV fluids
- Intestinal blockage or ileus requiring emergency treatment
- Chronic digestive problems persisting after stopping the drug
- Severe malnutrition or dehydration from inability to eat
- Need for feeding tube or IV nutrition (TPN)
Vision Injuries:
- Sudden vision loss (NAION) diagnosed by ophthalmologist
- Permanent or long-term vision impairment
- Eye stroke while taking GLP-1 medication
Other Serious Complications:
- Emergency surgery related to GI complications
- Multiple hospitalizations for medication side effects
- Gallbladder disease requiring surgery
- Pancreatitis confirmed by testing
Why Choose Our Firm?
Exclusive GLP-1 Focus We specialize specifically in GLP-1 injury cases. This isn’t a side practice - it’s what we do. Our attorneys have:
- Deep knowledge of the medications and their mechanisms
- Established relationships with top medical experts
- Experience with MDL 3094 procedures and requirements
- Track record of successful outcomes
Medical Expert Network We work with leading specialists who understand these injuries:
- Board-certified gastroenterologists who diagnose and treat gastroparesis
- Motility specialists who are experts in gastric emptying disorders
- Pharmacologists who can explain drug mechanisms and causation
- Ophthalmologists for vision loss cases
- Life care planners who calculate future medical needs
Proven Track Record Our firm has recovered millions of dollars in pharmaceutical MDL and complex litigation cases:
- Successfully represented clients in previous drug litigation
- Understand the bellwether trial process
- Know how to negotiate favorable settlements
- Won’t be intimidated by large pharmaceutical companies
No Upfront Costs We work on a contingency fee basis:
- No fee unless we win your case
- We advance all case costs
- Free initial consultation
- No obligation to proceed
- Transparent fee agreement
You risk nothing by finding out if you have a case.
Comprehensive Case Management We handle every aspect of your claim:
- Gathering all medical records
- Coordinating with your doctors
- Obtaining necessary diagnostic testing
- Working with medical experts
- Communicating with defendants
- Keeping you informed throughout
- Negotiating maximum compensation
Compassionate Support We understand these injuries are devastating:
- You’re not just a case number
- Direct attorney contact
- Responsive to your questions
- Respect for your situation
- Support through difficult process
Contact Us Today
Email: Info@GLP1Lawsuits.com
Online: Complete our free case evaluation form
Office Hours: Monday-Friday, 9:00 AM - 6:00 PM PT
Emergency Contact: Available for urgent matters
Next Steps After Contacting Us
Within 24 hours:
- An attorney or case manager will contact you
- Brief screening conversation
- Determine if you likely qualify
Additional Resources
Learn More About Your Rights:
- Do I Qualify? Signs You May Have a Case
- Understanding MDL 3094: What to Expect
- Medical Records Needed for Your Claim
- GLP-1 Lawsuit Criteria and Requirements
Understand the Injuries:
- Gastroparesis: Stomach Paralysis Explained
- Ileus and Bowel Obstruction Warning Signs
- Vision Loss from NAION: What You Need to Know
- Complete Guide to Gastric Emptying Scans
Understand the Medications:
- What Are GLP-1 Receptor Agonists?
- How GLP-1 Drugs Work: Mechanism Explained
- FDA Approval Timeline: What Regulators Knew
Frequently Asked Questions
“How much does it cost to find out if I have a case?” Nothing. Your initial consultation is completely free with no obligation.
“What if I’m not sure I have enough evidence?” That’s what we’re here to determine. We’ll review your situation and help gather any missing documentation.
“I’m still deciding whether to pursue legal action. Can I just talk to someone?” Absolutely. There’s no pressure. We’re happy to answer your questions and help you make an informed decision.
“How long does the legal process take?” Most MDL cases take 2-4 years from filing to resolution. Bellwether trials in mid-2026 will likely accelerate settlement discussions.
“Will I have to go to court?” Most cases settle without trial. If your case does go to trial, we’ll prepare you thoroughly and be with you every step.
“What if I took the medication a few years ago?” You may still be within the statute of limitations. Contact us immediately to check your deadline.
Take Action Today
Every day matters:
- Evidence may be lost over time
- Statutes of limitations are approaching for some patients
- Early filing may position you better in the MDL
- Your health and financial recovery are at stake
You have nothing to lose and potentially much to gain by finding out if you have a case.
Fill the online form now or email: Info@GLP1Lawsuits.com
Summary: Key Takeaways
The Medications Are Different, But the Risks Are Similar
Semaglutide drugs (Ozempic, Wegovy):
- Single GLP-1 agonist
- Novo Nordisk manufacturer
- Longer track record
- Most lawsuits filed
Tirzepatide drugs (Mounjaro, Zepbound):
- Dual GLP-1/GIP agonist
- Eli Lilly manufacturer
- Newer to market
- Growing litigation
All drugs:
- Slow gastric emptying (the dangerous mechanism)
- Can cause severe gastroparesis
- Associated with bowel obstruction, ileus, vision loss
- Subject to failure-to-warn lawsuits in MDL 3094
Legal Claims Apply Regardless of Which Drug You Took
- Gastroparesis claims require objective testing (gastric emptying scan)
- Both diabetes and weight-loss patients can file
- Multiple drug exposure doesn’t disqualify you
- Bellwether trials coming mid-2026
- Statute of limitations: 2-4 years depending on state
If You’ve Been Injured, Act Now
Time is critical:
- Statutes of limitations are running
- Evidence must be preserved
- Proper diagnostic testing required
- Early filing is advantageous
You deserve answers:
- Free case evaluation
- No cost unless we win
- Experienced attorneys
- Compassionate support
Don’t wait another day. Contact us now to protect your rights and pursue the compensation you deserve.
Disclaimer: This article is for informational purposes only and does not constitute legal or medical advice. Every case is unique. Consult with a qualified attorney to discuss your specific situation. Past results do not guarantee future outcomes.
Last reviewed by a licensed attorney: January 2026
© 2026 GLP1Lawsuits.com | All Rights Reserved