Sudden, permanent vision loss was never listed on the label for Ozempic, Wegovy, or other GLP‑1 drugs. But new research and safety alerts suggest it may be a real risk.
Mounting medical research and safety alerts now associate GLP‑1 drugs such as Ozempic, Wegovy, Mounjaro, and Zepbound with a rare but serious eye condition called non‑arteritic anterior ischemic optic neuropathy (NAION), which can cause sudden, often permanent vision loss in one or both eyes. At the same time, a new federal multidistrict litigation (MDL) has been created specifically for GLP‑1 vision‑loss and NAION claims, separate from the existing GLP‑1 MDL focused on gastrointestinal injuries.
This article explains what NAION is, how GLP‑1 drugs may be linked to optic‑nerve damage, current medical and regulatory guidance, and what these developments may mean for potential NAION vision‑loss lawsuits.
What Are GLP‑1 Drugs?
Glucagon‑like peptide‑1 receptor agonists (GLP‑1 RAs) are injectable or oral medications originally developed to treat type 2 diabetes and later approved or widely used off‑label for weight loss. Popular brand‑name GLP‑1 drugs include:
- Ozempic and Wegovy (semaglutide)
- Mounjaro and Zepbound (tirzepatide)
- Saxenda and Victoza (liraglutide)
- Trulicity (dulaglutide)
- Rybelsus (oral semaglutide)
These drugs improve blood‑sugar control, promote weight loss, and reduce cardiovascular risk for many patients, which is why their use has surged in recent years. However, as prescriptions have increased, doctors and regulators have also begun tracking rare but serious side effects, including eye complications such as NAION.
What Is NAION and Why Does It Matter?
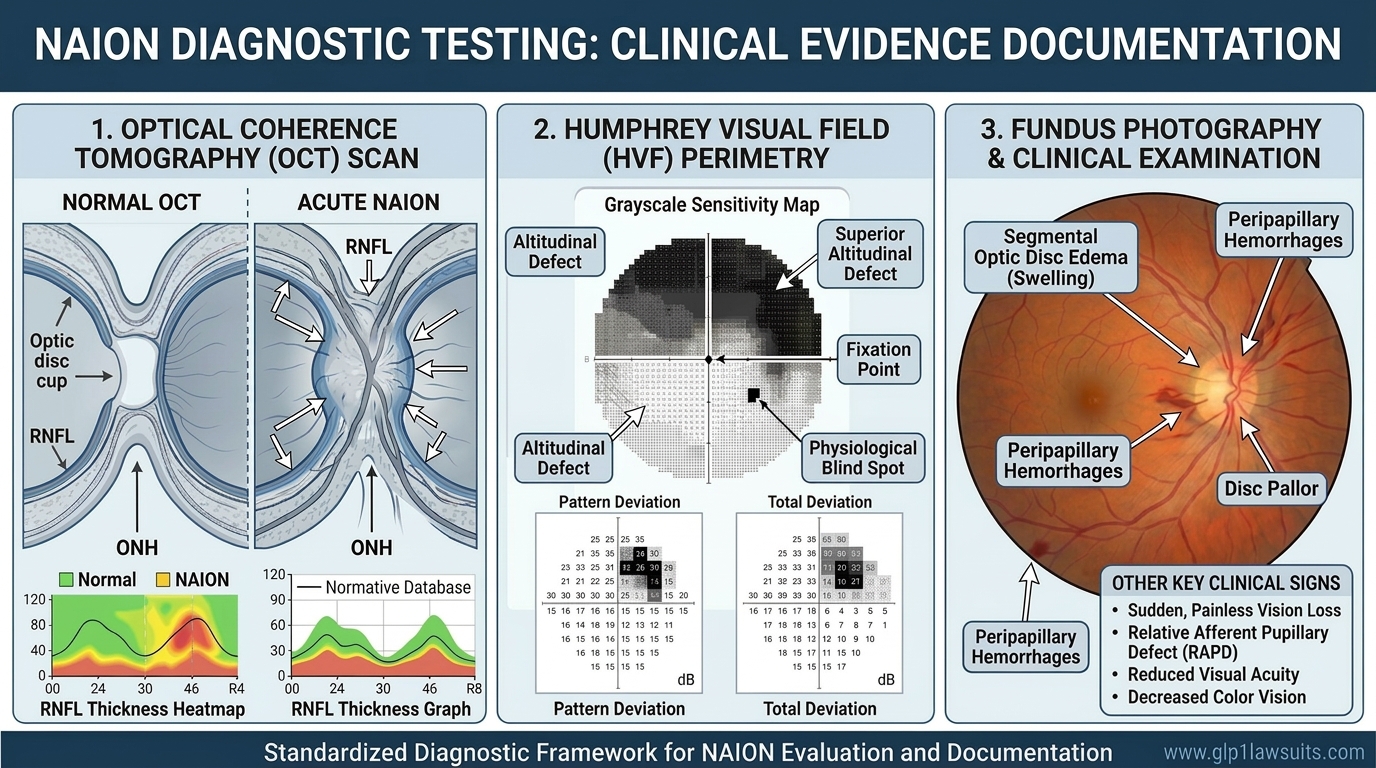
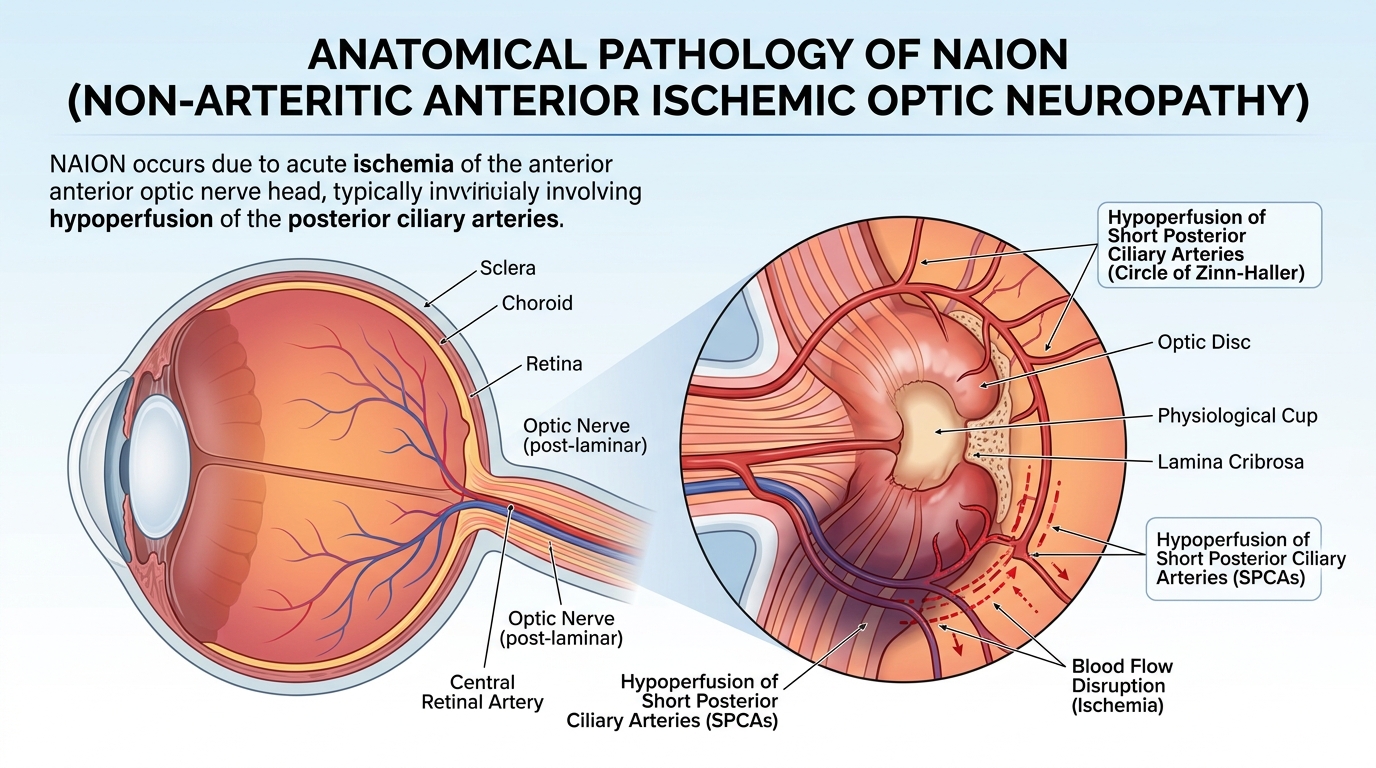
NAION (non‑arteritic anterior ischemic optic neuropathy) is a form of optic‑nerve stroke caused by reduced blood flow to the front portion of the optic nerve. It typically presents as sudden, painless vision loss in one eye, often noticed upon waking, along with visual‑field defects, color‑vision changes, and optic‑disc swelling on exam.
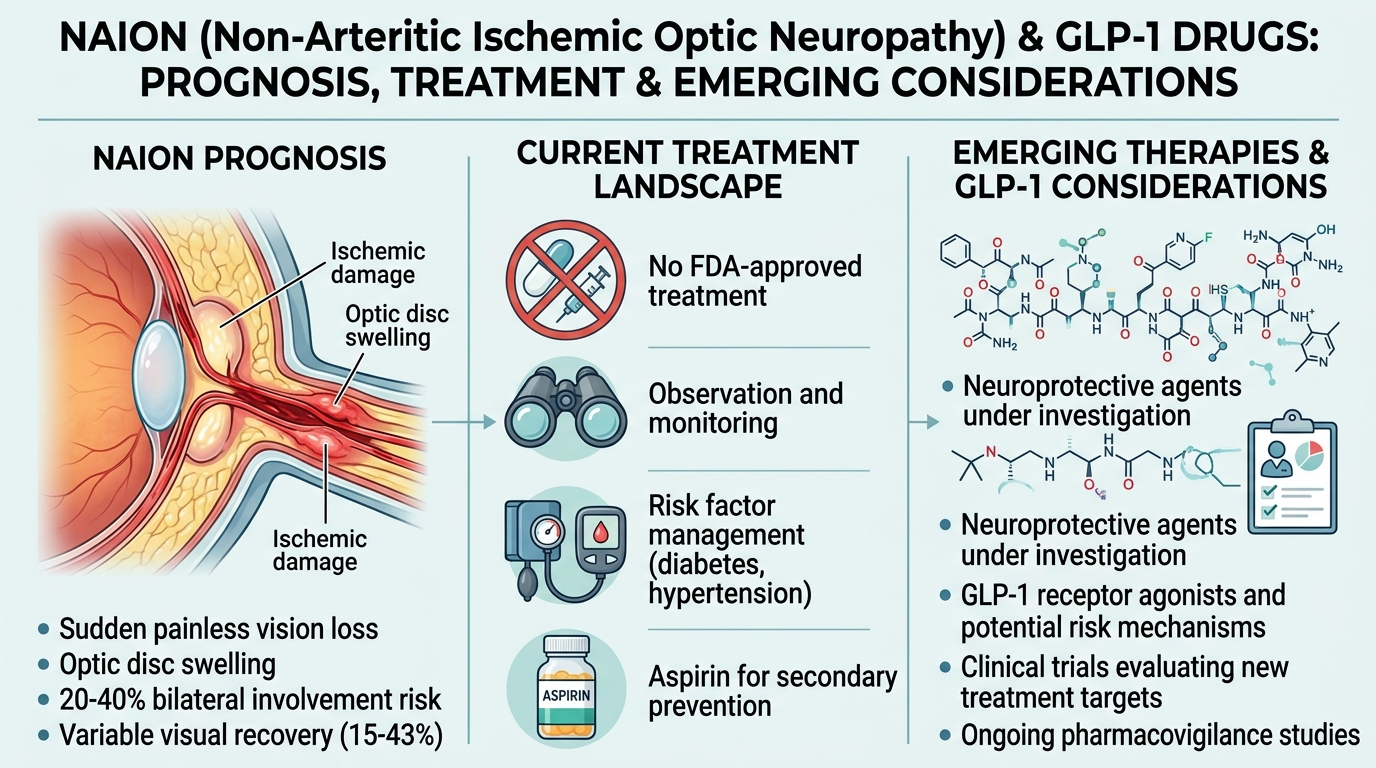
NAION is one of the most common causes of permanent vision loss in adults and is the second most common optic neuropathy after glaucoma. There is currently no proven treatment that reliably restores lost vision; management focuses on addressing risk factors and preventing further damage.
For GLP‑1 users, the concern is not that NAION is common, but that even a very low risk can be devastating because vision loss is often irreversible and life‑altering. Even mild NAION can permanently impair driving, reading, and depth perception.
Emerging Research Linking GLP‑1 Drugs and NAION
Semaglutide (Ozempic, Wegovy) and NAION
A matched‑cohort study of 16,827 patients published in JAMA Ophthalmology found a significantly higher risk of NAION among patients prescribed semaglutide compared with similar patients prescribed non‑GLP‑1 antidiabetic medications. NAION occurred more than seven times as often in semaglutide users as in the comparison group, though the absolute number of cases remained small.
A Danish nationwide study followed more than 400,000 patients with type 2 diabetes and reported that weekly semaglutide use was associated with more than double the risk of NAION compared with other diabetes drugs. Additional case reports have described individual patients developing NAION after starting semaglutide, with symptom onset temporally related to the medication. Although researchers emphasize that the overall risk remains low, multiple independent datasets now point to a consistent safety signal across semaglutide and tirzepatide users.
Semaglutide or Tirzepatide and Optic‑Nerve Disorders
A large target‑trial emulation using data from approximately 1.5 million patients with type 2 diabetes compared semaglutide or tirzepatide to other antidiabetic medications. The study found that semaglutide or tirzepatide was associated with increased risk of NAION and other optic‑nerve disorders, although the overall absolute risk remained low and there was no increased risk for conditions such as optic neuritis or papilledema.
A 2026 cohort analysis similarly concluded that GLP‑1 therapy, particularly newer agents such as semaglutide and tirzepatide, was associated with a higher incidence of NAION over five years, reinforcing concerns about a real but rare safety signal.
Professional and Regulatory Perspectives on the Evidence
The American Optometric Association (AOA) has issued guidance noting that GLP‑1 RAs are “very positive and transformative” medications but acknowledging rare ocular complications, including NAION, which can cause permanent vision loss. The AOA’s report cites data suggesting a roughly two‑fold increased NAION risk with semaglutide and estimates that NAION may affect up to 1 in 10,000 semaglutide users.
Regulators at the European Medicines Agency (EMA) and the World Health Organization (WHO) have reviewed global safety data and concluded that NAION is a “very rare” side effect of semaglutide, recommending label updates and urgent evaluation for any sudden vision changes. While some ophthalmology experts emphasize that GLP‑1 benefits still outweigh the small NAION risk, they also call for baseline eye exams and close monitoring, especially in high‑risk patients.
How Rare Is NAION in GLP‑1 Users?
NAION remains an uncommon complication, even among GLP‑1 users, but the severity of potential harm has drawn intense scrutiny. The EMA’s safety committee and WHO estimate that NAION may occur in up to 1 in 10,000 semaglutide users, categorizing it as a very rare adverse effect.
Real‑world epidemiologic studies suggest that patients on semaglutide or other GLP‑1 agents may have roughly a two‑ to three‑fold increased risk of NAION compared with patients on other diabetes medications, though precise risk varies by study and population. Even with this relative increase, the absolute number of cases is still small compared with the millions of people receiving GLP‑1 prescriptions. For context, this risk level is significantly higher than what is traditionally expected in the general population.
For an individual patient, this means the chance of NAION is low. But if it occurs, the resulting vision loss may be permanent and disabling.
Who May Be at Higher Risk for NAION?
NAION typically occurs in people with underlying vascular or structural risk factors, many of which overlap with the reasons patients are prescribed GLP‑1 drugs in the first place. Key risk factors include:
- A “crowded” or small optic‑disc cup (an anatomical variant strongly associated with NAION)
- Diabetes, hypertension, and high cholesterol
- Obstructive sleep apnea and nocturnal hypotension (low blood pressure at night)
- Older age and other cardiovascular disease risk factors
- Rapid dose escalation, which some GLP‑1 prescribers use for weight‑loss titration, may further stress optic‑nerve perfusion in susceptible patients
Experts have hypothesized that rapid weight loss, fast improvements in blood sugar, and GLP‑1‑related effects on vascular tone may combine with these pre‑existing factors to reduce blood flow to a structurally vulnerable optic nerve, triggering NAION in susceptible individuals. Because many GLP‑1 patients already have diabetes, hypertension, or sleep apnea, they may be in a higher‑baseline‑risk group even before starting these medications.
Warning Signs of NAION and When to Seek Emergency Care
NAION is considered an ophthalmic emergency because early evaluation is crucial to rule out other causes and to document findings that may support future treatment or legal claims. Common warning signs include:
- Sudden, painless vision loss in one eye, often noticed upon waking
- Blurred or foggy vision, dark or shadowy areas, or a “curtain” across part of the visual field
- New blind spots or loss of peripheral or upper/lower half of vision (altitudinal defect)
- Loss or dulling of color vision (dyschromatopsia)
- A relative afferent pupillary defect and optic‑disc swelling when examined by an eye doctor
If you experience sudden vision changes while taking Ozempic, Wegovy, Mounjaro, Zepbound, or another GLP‑1 medication, medical organizations and regulators advise seeking urgent ophthalmologic evaluation and notifying your prescriber immediately. The EMA and WHO recommend discontinuing semaglutide if NAION is confirmed.
📖 For a deeper dive into red‑flag symptoms and when to treat vision loss as an emergency, read our dedicated article: Sudden Vision Loss: When to Seek Emergency Care
What Do Medical Societies and Regulators Say Now?
The American Academy of Ophthalmology and related expert reviews note that GLP‑1 drugs remain valuable treatments and are not being broadly withdrawn from the market, but that clinicians should be aware of the potential NAION risk. Existing guidance emphasizes baseline and follow‑up eye exams for GLP‑1 users, individualized risk counseling, and close coordination between eye doctors and the clinicians managing diabetes and cardiovascular health.
The AOA’s Evidence‑Based Optometry Committee recommends that patients on GLP‑1 RAs be considered “at‑risk,” with more frequent re‑examinations and prompt referral if visual symptoms develop. The EMA and WHO have gone further by formally recognizing NAION as a very rare side effect of semaglutide, updating risk‑management plans, and directing manufacturers to add stronger warnings.
📖 Our articles will explore these positions in more detail:
Current Understanding of Prognosis and Treatment
Despite active research, there is still no widely accepted treatment that can reliably reverse vision loss once NAION has occurred. Some small studies suggest that steroids may offer limited visual benefit for certain patients, but data are inconclusive and must be weighed against risks such as worsened blood‑sugar control.
Most guidelines focus on eliminating potential triggers (including discontinuing the suspected medication if NAION is diagnosed), optimizing cardiovascular risk factors, and monitoring the fellow eye—which faces an increased risk of later NAION. Because NAION is often permanent, realistic expectations about recovery and long‑term adaptation are crucial.
📖 We will address these issues in more depth in:
NAION and GLP‑1 Vision‑Loss Lawsuits
As scientific evidence and safety alerts have accumulated, litigation involving GLP‑1 drugs has expanded beyond gastrointestinal injuries to include vision‑loss claims. Plaintiffs allege that manufacturers such as Novo Nordisk and Eli Lilly failed to adequately warn about the risk of NAION and irreversible optic‑nerve damage associated with medications including Ozempic, Wegovy, Mounjaro, and related GLP‑1 products.
A new federal MDL No. 3163 has been established specifically for GLP‑1 vision‑loss and NAION claims, recognizing that these injuries raise distinct medical and legal questions from the existing GLP‑1 MDL No. 3094, which focuses on gastroparesis and other gastrointestinal side effects. In addition to the federal MDL, there are efforts to consolidate NAION lawsuits at the state level, including multicounty litigation petitions in jurisdictions such as New Jersey. NAION vision‑loss lawsuits now form a distinct category within the broader GLP‑1 litigation landscape.
What to Do If You Had Vision Loss After GLP‑1 Use
If you experienced sudden vision changes or were diagnosed with NAION after using a GLP‑1 drug, medical and legal experts generally recommend the following steps:
- Seek immediate evaluation by an ophthalmologist or neuro‑ophthalmologist to document findings, confirm the diagnosis, and rule out other causes.
- Inform your prescriber who manages your GLP‑1 therapy and discuss whether the medication should be stopped, in line with EMA and WHO recommendations for confirmed NAION.
- Preserve medical records, imaging (such as OCT and visual‑field tests), and pharmacy records linking your GLP‑1 prescriptions to the timing of vision loss.
- Consult an experienced pharmaceutical‑litigation attorney to review your situation, explain the current MDL landscape, and determine whether you may have a viable NAION vision‑loss claim.
📖 Our articles will walk through the specific diagnostics important both medically and legally:
Q&A: GLP‑1 Drugs, NAION, and Lawsuits
Q1. Are GLP‑1 drugs causing blindness?
Studies do not show that GLP‑1 drugs cause blindness in most users, but they do indicate a significantly increased risk of NAION and other optic‑nerve disorders compared with some other diabetes medications, while the overall risk remains low. Because NAION often leads to permanent vision loss in one eye, even this rare risk is taken very seriously by ophthalmologists and regulators.
Q2. How can I tell if my vision loss might be NAION?
NAION typically presents as sudden, painless vision loss in one eye, usually noticed upon waking, accompanied by blurred or foggy vision, darkened or shadowy areas, and sometimes loss of color vision. Only a comprehensive eye exam, including dilated fundus evaluation and often visual‑field testing and imaging, can confirm NAION and distinguish it from other causes such as retinal detachment or stroke.
Q3. Should I stop my GLP‑1 medication if I am worried about NAION?
Professional societies currently do not recommend that all patients stop GLP‑1 therapy solely due to NAION concerns, emphasizing that benefits still generally outweigh the very small absolute risk. However, regulators advise that semaglutide should be discontinued if NAION is confirmed, and any new vision symptoms while on GLP‑1 treatment warrant urgent eye evaluation and prompt discussion with your prescribing doctor.
Q4. Can lost vision from NAION be reversed?
Most studies and reviews conclude that there is no proven treatment that reliably restores vision once NAION has occurred, although some patients experience partial spontaneous improvement over time. Management focuses on stabilizing the condition, addressing systemic risk factors, and monitoring the unaffected eye, which may also be at higher risk.
Q5. Do I qualify for a NAION vision‑loss lawsuit?
Potential factors supporting a NAION claim may include: documented use of a GLP‑1 medication (such as Ozempic, Wegovy, Mounjaro, or similar drugs), a confirmed NAION diagnosis or sudden vision loss consistent with NAION, and medical records linking the timing of symptoms with GLP‑1 use. Only a licensed attorney can evaluate how these factors apply to your situation and whether your case may be eligible for inclusion in the GLP‑1 NAION MDL or related proceedings.
Q6. Can vision loss happen in both eyes?
While NAION typically begins in one eye, approximately 15–24% of patients experience NAION in the other eye within five years, especially when risk factors remain unaddressed.
Why Work With Us
Exclusive Focus on GLP‑1 Injuries
Our practice is dedicated entirely to GLP‑1 medication injuries—including NAION vision loss, gastroparesis, ileus, and related complications. This is not a side project or add‑on; it is the core of what we do every day.
- Deep, case‑specific knowledge of Ozempic, Wegovy, Mounjaro, Zepbound, and related medications
- Familiarity with how these drugs work, their known side‑effect profiles, and the evolving medical literature around NAION and other serious harms
- Hands‑on experience with MDL No. 3094 & MDL No 3163 procedures, case‑management orders, and plaintiff requirements in complex pharmaceutical litigation
Because we live in this space, we can quickly identify the strengths and weaknesses of a potential GLP‑1 claim and tailor your case strategy around the most up‑to‑date science and MDL developments.
Integrated Medical Expert Network
GLP‑1 and NAION cases are built on medicine as much as law. We work closely with a network of highly qualified specialists who understand these specific injuries:
- Board‑certified gastroenterologists and motility specialists for gastroparesis and gastric‑emptying disorders
- Pharmacologists who can explain GLP‑1 mechanisms, dosing, and causation theories in plain language for judges and juries
- Ophthalmologists and neuro‑ophthalmologists for NAION and other optic‑nerve injuries
- Life‑care planners and vocational experts who quantify future medical needs, work limitations, and long‑term costs
This multidisciplinary team helps us document what happened medically, how it connects to the drug, and what you will likely face in the future, key issues in both settlement negotiations and trial.
Proven Results in Complex Drug Litigation
Our attorneys have collectively recovered millions of dollars for clients in pharmaceutical MDLs and other complex injury cases, including claims against major drug manufacturers. While past results cannot guarantee future outcomes, our experience shows that we:
- Know how to develop cases for bellwether trials and global‑resolution talks
- Understand how to work within MDL structures while preserving your individual story and damages
- Are prepared to stand up to large pharmaceutical companies and their insurers in discovery, motion practice, and negotiation
Our goal in GLP‑1 litigation is straightforward: to position your claim for the strongest possible recovery under the facts and the law.
No Upfront Costs – Ever
We represent GLP‑1 clients on a contingency‑fee basis:
- No attorney fee unless we obtain a recovery for you
- We advance the costs of experts, records, and litigation
- Free, no‑obligation initial consultation
- Clear, written fee agreement from the beginning
You can find out whether you have a potential GLP‑1 or NAION claim without risking a single dollar of your own money.
Comprehensive Case Management
GLP‑1 injury and NAION cases demand detailed documentation. We take on the heavy lifting so you can focus on your health:
- Collecting and organizing medical records, pharmacy data, imaging, and diagnostic test results
- Coordinating with your treating doctors and consulting specialists
- Ensuring you obtain critical diagnostic testing tied to NAION and other alleged GLP‑1 injuries
- Preparing and submitting required MDL forms, plaintiff fact sheets, and authorizations
- Communicating with defendants and insurers and keeping you updated at each major step
- Pursuing maximum compensation for medical bills, lost income, pain and suffering, and future care needs
From intake to resolution, we work to make a complex process as manageable and transparent as possible.
Compassionate, Client‑Centered Support
We understand that sudden vision loss, chronic gastroparesis, and other GLP‑1‑related injuries are life‑changing, not just “cases.” You can expect:
- Direct access to your legal team, not just a call center
- Prompt responses to questions and clear explanations at every stage
- Respect for your circumstances, schedule, and comfort level
- Guidance and support through medical, financial, and legal uncertainty
Our team works with MDL Steering Committee counsel and other leadership where appropriate, so your case benefits from the broader discovery record and litigation strategy while still receiving individualized attention.
Contact Us for a Free Consultation
Vision loss is permanent, but your legal rights are not. Filing deadlines apply.
If you took Ozempic, Wegovy, Mounjaro, Zepbound, Trulicity, or other GLP-1 drugs and developed sudden vision changes, suspected NAION, or documented optic‑nerve damage, you may have a legal claim against the manufacturers of these GLP‑1 medications. Our team follows the latest medical evidence and court developments in the emerging GLP‑1 NAION vision‑loss MDL and related state‑court proceedings.
Visit www.GLP1Lawsuits.com to:
- Get a free case evaluation
- Speak with an experienced pharmaceutical‑litigation attorney about NAION and GLP‑1 drugs
- Learn more about your legal options in federal MDL proceedings and related state‑court consolidations
- Obtain timeline‑specific analysis of your GLP‑1 use, diagnosis, and vision‑loss history
- Join others seeking justice and accountability for preventable optic‑nerve injuries
Do not wait until the statute of limitations runs out. Courts handling GLP‑1 litigation apply strict filing deadlines that can permanently bar otherwise valid claims if missed. Contact us today to protect your rights and pursue the compensation you deserve - no upfront fees and no costs unless we win your case.
Sources Cited
| # | Authority | Description |
|---|---|---|
| 1 | American Optometric Association (AOA) | Clinical report on GLP‑1 receptor agonists and ocular health; public guidance on NAION risk in GLP‑1 users (2025) |
| 2 | World Health Organization (WHO) | Global safety communication: “The use of semaglutide medicines and risk of NAION” (June 2025) |
| 3 | European Medicines Agency (EMA) / PRAC | Pharmacovigilance conclusion: NAION classified as a very rare side effect of semaglutide; label‑update directive (2025) |
| 4 | JAMA Ophthalmology | Matched‑cohort study of 16,827 patients: “Risk of Nonarteritic Anterior Ischemic Optic Neuropathy in Patients Prescribed Semaglutide” (2024) |
| 5 | JAMA Network Open | Large target‑trial emulation (~1.5 million patients): “Semaglutide or Tirzepatide and Optic Nerve and Visual Pathway Disorders in Type 2 Diabetes” (2025) |
| 6 | NIH / PubMed Central (PMC) | Peer‑reviewed reviews and case reports on GLP‑1 drugs and NAION, including: Sight Unseen: GLP‑1 Agonism Therapy and the Eye (2026); individual NAION case reports following semaglutide initiation |
| 7 | Judicial Panel on Multidistrict Litigation (JPML) – MDL No. 3094 | Transfer orders centralizing GLP‑1 RA Products Liability Litigation; subsequent orders on MDL scope and structure (2024) |
Disclaimer
This article provides general information about GLP‑1 medications, NAION, and related litigation and does not constitute medical or legal advice. For diagnosis, treatment, or questions about your medications, consult a qualified healthcare provider such as an ophthalmologist, endocrinologist, or primary‑care physician. For legal guidance, consult a licensed attorney who can evaluate your specific facts, including medical records, prescription history, and applicable deadlines. Each case is unique, and past results do not guarantee future outcomes.
© 2026 GLP1lawsuits.com | All Rights Reserved
Last reviewed by a licensed attorney: March, 2026