Does the science prove GLP-1 drugs cause NAION? Here is what the full body of evidence, eight major studies, two regulatory agencies, a global pharmacovigilance database of 30 million reports, and the Bradford Hill causation framework, actually says.
Of all the articles in this series, this is the one that matters most in a courtroom. When a jury, a federal judge ruling on Daubert challenges, or opposing counsel evaluating a settlement hears the words “GLP-1 drugs caused NAION,” the question that immediately follows is: How do you know?
The answer to that question is built from epidemiology, pharmacology, vascular biology, regulatory science, and classical causation analysis. This article assembles all of it, clearly, honestly, and with enough scientific depth to serve as a reference for both patients and the attorneys representing them.
For a patient-facing introduction to the GLP-1/NAION link, read: Understanding the Growing Concerns Around GLP-1 Drugs and Vision Loss (NAION) For what NAION is and how it damages the optic nerve, read: What Is NAION? Understanding Non-Arteritic Anterior Ischemic Optic Neuropathy For the litigation status of NAION claims, read: NAION MDL Update: GLP-1 Vision Loss Litigation Status
Part I: The Science of Why Diabetes Already Sets the Stage for NAION
Before analyzing the GLP-1 drug contribution, it is essential to understand what diabetes does to the optic nerve, because the two risk factors are intertwined in a way that is both medically important and legally significant.
Diabetes as an Independent NAION Risk Factor
Diabetes mellitus is one of the most consistently documented systemic risk factors for NAION. The mechanisms are multiple and converging:
1. Optic nerve head microvascular disease: Diabetes causes small-vessel disease throughout the body, including the capillaries and short posterior ciliary arteries that supply the optic nerve head. Chronic hyperglycemia thickens basement membranes, damages endothelial cells, increases blood viscosity, and impairs autoregulation of optic nerve perfusion. Studies have found reduced optic nerve head blood flow in diabetic patients even before NAION occurs.
2. Hypercoagulability: Patients with type 2 diabetes have a well-documented increased tendency for blood clot formation, elevated platelet activation, increased fibrinogen, and impaired fibrinolysis, all of which increase the risk of microvascular occlusion in the optic nerve head.
3. Disc-at-risk amplification: Diabetes does not cause the “disc-at-risk” anatomy (a small, crowded optic disc with minimal cupping), but it dramatically amplifies the clinical consequence of having it. A patient with a disc-at-risk and controlled blood sugar may never develop NAION; the same patient with poorly controlled diabetes, with its attendant microvascular disease and reduced perfusion reserve, faces significantly higher risk.
4. Fellow-eye vulnerability: A 2025 study published in PMC demonstrated that diabetic patients with NAION in one eye had a median of just 14 months before developing NAION in the fellow eye, compared with 122 months in normoglycemic patients. This eightfold compression of the fellow-eye attack interval illustrates how profoundly diabetes accelerates optic nerve vulnerability.
5. Dyslipidemia and LDL: A 2025 multivariable logistic regression study found that dyslipidemia (OR=8.36) and elevated LDL (OR=1.017 per unit) were the strongest independent vascular risk factors for NAION in type 2 diabetes patients, even stronger than blood pressure alone. Since dyslipidemia and type 2 diabetes are frequently co-occurring and are the very conditions GLP-1 drugs are prescribed to manage, this creates a layered risk picture.
What This Means for Causation Analysis
The fact that diabetes independently increases NAION risk does not exonerate GLP-1 drugs. It creates the biological context in which GLP-1 drugs may act as an additional insult, the “final hit” on an already vulnerable optic nerve. In litigation, the defense will argue that diabetes alone caused the plaintiff’s NAION; plaintiffs’ experts must explain the drug’s additive or independent contribution using the mechanistic and epidemiological evidence reviewed below.
Part II: The Epidemiological Evidence: Eight Major Studies
The scientific literature on GLP-1 drugs and NAION has grown rapidly since the first major study was published in July 2024. Here is a comprehensive review of every significant study published through early 2026, with key data presented precisely.
Study 1: Hathaway et al. JAMA Ophthalmology (July 2024)
The study that launched the litigation
- Design: Matched retrospective cohort study at Massachusetts Eye and Ear (Harvard Medical School)
- Population: 16,827 patients total, semaglutide vs. non-GLP-1 antidiabetic medications (diabetes cohort); semaglutide vs. non-GLP-1 weight-loss medications (obesity cohort)
-
Key findings:
- Diabetes cohort: HR 4.28 (95% CI 1.62–11.29), semaglutide users were more than 4× as likely to develop NAION
- Obesity cohort: HR 7.64 (95% CI 2.21–26.36), semaglutide users were more than 7× as likely to develop NAION
- Temporal association: elevated risk observed within the first year of semaglutide initiation
-
Why it matters: This was the first large, peer-reviewed, published study specifically designed to test the NAION-semaglutide association. Its publication in JAMA Ophthalmology, among the most prestigious journals in its field, and its use of propensity-score matching to control for confounders gave it immediate scientific and legal significance.
Study 2: Danish Nationwide Cohort (2024)
The largest national real-world population study
- Design: Nationwide population-based cohort study using Danish health registries
- Population: 424,152 patients with type 2 diabetes, the largest single-nation cohort studied
- Key finding: Semaglutide was associated with an increased risk of NAION: HR 2.19 (95% CI 1.54–3.12)
- Why it matters: Denmark’s comprehensive national health registry allows for complete population-level follow-up with minimal selection bias. A hazard ratio of 2.19 from over 400,000 patients is among the strongest large-scale confirmatory evidence available and directly addresses consistency concerns across different populations.
Study 3: Semaglutide or Tirzepatide and Optic Nerve Disorders Cohort
Published in JAMA Network Open (2025), extending findings to tirzepatide
- Design: Propensity-score matched retrospective cohort
- Population: 159,398 matched patients with type 2 diabetes (79,699 per cohort), semaglutide or tirzepatide vs. other antidiabetic medications
-
Key findings:
- NAION incidence: 35 patients (0.04%) in semaglutide/tirzepatide group vs. 19 patients (0.02%) in comparison group
- HR 1.76 (95% CI 1.01–3.07), 76% relative increase in NAION risk
- Cumulative incidence curves began diverging immediately after medication initiation and separated throughout the follow-up period, a critical temporal finding
- The elevated risk was observed for both semaglutide and tirzepatide, though individual drug analyses were limited by sample size
-
Why it matters: This study extends the association beyond semaglutide to tirzepatide (Mounjaro, Zepbound), broadening the scope of litigation. The diverging cumulative incidence curves from day one are particularly powerful evidence of a temporally consistent drug-related effect.
Study 4: Five-Year Retrospective Cohort, PMC (2026)
The longest follow-up period in the literature
- Design: Propensity-score matched retrospective cohort; 5-year follow-up
- Population: 776,666 patients total (388,333 per cohort), the largest study by patient count
-
Key findings:
- Risk difference: 0.022% (95% CI 0.01%–0.034%)
- Risk ratio: 1.339 (95% CI 1.137–1.577; p=0.005), statistically significant
- Absolute 5-year risk in GLP-1 users: 0.087%
- E-value sensitivity analysis confirmed moderately robust association, meaning the association could only be explained by an unmeasured confounder with a risk ratio of approximately 2.0 for both exposure and outcome
-
Why it matters: The 5-year follow-up is the longest window studied, confirming that the elevated risk is not limited to the first year but persists throughout extended GLP-1 use. The E-value analysis strengthens causal inference by quantifying how large an unmeasured confounding effect would need to be to explain away the association.
Study 5: Large Observational Study: GLP-1RA vs. SGLT2 Inhibitors (Published 2026)
Meta-analysis confirms 85% increased risk; pooled HR 2.78
- Design: Large observational study + meta-analysis combining new results with previously published data
- Comparator: SGLT2 inhibitors (another major class of antidiabetic medication), a particularly clean comparator because SGLT2i users have similar diabetes severity and cardiovascular risk profiles
-
Key findings:
- New study: HR 1.85 (85% increased risk of presumed NAION vs. SGLT2 initiators)
- Meta-analysis combining this study with prior results: HR 2.78 (95% CI 1.39–5.56)
- Results were consistent across subgroups and sensitivity analyses
-
Why it matters: Using SGLT2 inhibitors as a comparator rather than non-medicated patients eliminates a major confounding concern that GLP-1 users simply have worse baseline health. When compared with similarly diabetic patients on a different drug class, GLP-1 users still show significantly elevated NAION risk. The meta-analytic pooled HR of 2.78 represents the current best summary estimate of the relative risk from the published observational literature.
Study 6: FDA FAERS Pharmacovigilance Analysis (2025)
30 million reports, formulation-specific and sex-stratified findings
- Design: Disproportionality analysis using the FDA Adverse Event Reporting System (FAERS), over 30 million adverse event reports (2017–2024); among 31,774 semaglutide cases analyzed
-
Key findings:
- Wegovy (weight-loss semaglutide) ROR: 74.89 for ischemic optic neuropathy, an extraordinarily strong signal
- Ozempic (diabetes semaglutide) ROR: 18.81
- Sex-stratified: Men showed higher signal (ROR 116.37 in men vs. lower in women)
- Multivariable regression confirmed Wegovy vs. Ozempic: adjusted OR 4.74; men vs. women: adjusted OR 3.33
- The Wegovy-specific signal suggests a potential dose-dependent effect, Wegovy is prescribed at higher doses (up to 2.4 mg/week) than Ozempic (up to 1.0–2.0 mg/week for diabetes)
-
Why it matters: FAERS reporting odds ratios are a standard pharmacovigilance tool for detecting drug safety signals. An ROR of 74.89 for Wegovy, meaning that among all adverse event reports for all drugs in the database, ischemic optic neuropathy was reported at nearly 75× the expected rate for Wegovy, is one of the strongest signals in the GLP-1/NAION literature. This data directly supports the dose-response Bradford Hill criterion (see Part IV). The sex-specific finding also introduces a new dimension to individual plaintiff profiling.
Study 7: WHO Global Pharmacovigilance Database: AAO 2025
WHO data: 68.6× increased NAION rate vs. comparators
- Design: Analysis of the WHO’s global database of drug safety reports; 117,173 diabetic patients on semaglutide, empagliflozin, exenatide, tirzepatide, or insulin
- Key finding: Semaglutide users were 68.6 times more likely to develop NAION than those taking empagliflozin, exenatide, insulin, or metformin, presented at the American Academy of Ophthalmology 2025 annual meeting
- Why it matters: Global real-world pharmacovigilance data from WHO represents the broadest possible capture of NAION signals across diverse populations, prescribing systems, and medical traditions. A 68.6× reporting rate is an exceptional signal by any pharmacovigilance benchmark and extends the evidence beyond U.S. populations.
Study 8: Nature: Bradford Hill Pharmacovigilance Analysis (2025)
The study that explicitly applies Bradford Hill criteria to GLP-1/NAION
- Design: Pharmacovigilance analysis of neurological adverse events including NAION, with explicit application of Bradford Hill’s causation criteria
- Key finding: Concluded that as per Bradford Hill’s criteria, biological plausibility is supported through GLP-1 receptor expression in the nervous system and temporal consistency in time-to-event analyses, two of the most important causation criteria
- Why it matters: This is the study most directly relevant to Daubert proceedings, it explicitly frames the GLP-1/NAION association within the causation framework courts use to evaluate expert testimony. We apply this framework in full in Part IV below.
Where the Science Is Not Unanimous: Studies Finding No Increased Risk
Scientific credibility requires acknowledging the full literature, including studies that did not confirm the association:
- FDA Sentinel Initiative Analysis (September 2025): A large study using the FDA’s Sentinel network found no statistically significant increased risk of NAION with GLP-1 use. This analysis has been noted by defense experts in MDL proceedings and will be a central battleground in Daubert hearings.
- Review of Optometry / Newer Study (March 2025): One semaglutide study found no increased NAION risk, attributed in part to methodological differences including comparator selection and NAION ascertainment methods.
How plaintiffs’ experts address these diverging results: The scientific debate is not unusual in emerging drug safety litigation; it mirrors early contested studies in cigarette-lung cancer, statins-muscle injury, and SSRI-birth defect litigation. The weight of the evidence, particularly the meta-analytic pooled HR of 2.78, the FAERS ROR of 74.89 for Wegovy, the WHO signal of 68.6×, and the EMA/WHO formal regulatory recognition, is substantially on the side of an elevated risk. Plaintiffs’ general causation experts are expected to argue that the conflicting studies reflect methodological differences (particularly in how NAION is identified and which comparators are used) rather than true null effects.
Part III: The Biological Mechanisms: How GLP-1 Drugs May Damage the Optic Nerve
Epidemiological associations gain enormous scientific weight when a plausible biological mechanism explains them. In the GLP-1/NAION context, researchers have proposed several interacting mechanisms, none conclusively proven yet, but collectively forming a coherent and biologically plausible causal story.
Mechanism 1: Rapid Glucose Lowering and Osmotic Optic Nerve Stress
GLP-1 drugs are highly effective at reducing blood glucose, often achieving rapid, sustained reductions in HbA1c that occur within weeks to months of initiation. In a patient with chronic hyperglycemia, the optic nerve head tissue has equilibrated to the elevated glucose environment over months or years. When blood glucose drops rapidly:
- Intravascular osmotic pressure falls faster than extravascular tissues can equilibrate
- This creates an osmotic gradient that draws fluid from the intravascular space into the optic nerve tissue, generating swelling within the already-crowded optic nerve canal
- This swelling raises pressure within the scleral canal that surrounds the optic nerve head, compressing the tiny capillaries that supply the nerve, a classic compartment syndrome mechanism
- Rapid glucose correction has a well-documented analog: “insulin neuritis,” where rapid normalization of hyperglycemia with insulin causes optic nerve edema and, in extreme cases, infarction. GLP-1 drugs may trigger the same mechanism through a different pharmacological route.
Legal significance: The osmotic mechanism provides an explanation for why NAION risk appears elevated particularly early in GLP-1 treatment, the period of most rapid glucose reduction, consistent with the observation in the Hathaway study that temporal association was most prominent in the first year.
Mechanism 2: GLP-1 Receptor-Mediated Vascular Dysregulation
GLP-1 receptors are expressed not only in the pancreas but throughout the nervous system, cardiovascular system, and, critically, in the blood vessels and ganglion cells of the eye. GLP-1 receptor activation has multiple vascular effects that may be beneficial in most tissues but potentially harmful in the structurally vulnerable optic nerve head:
- Vasodilation: GLP-1 receptor agonism causes vasodilation in multiple vascular beds. In most patients this is protective, reducing cardiovascular events. In the optic nerve head, however, vasodilation in the feeding vessels may paradoxically reduce perfusion pressure if systemic blood pressure simultaneously drops (as it often does with weight loss and improved blood sugar control)
- Nocturnal hypotension amplification: GLP-1 drugs promote weight loss and blood pressure reduction. Patients with NAION risk factors already tend toward nocturnal hypotension, a drop in blood pressure during sleep that reduces optic nerve head perfusion. GLP-1-driven blood pressure reduction may amplify this nocturnal dip, reducing optic nerve head perfusion during the vulnerable sleeping hours, consistent with NAION’s classic morning-onset presentation
- Plasma volume contraction: Rapid weight loss and improved glycemic control reduce circulating plasma volume. Reduced plasma volume decreases the effective perfusion pressure to end-organ capillary beds including the optic nerve head
Legal significance: The vascular dysregulation mechanisms explain why higher-dose Wegovy (weight-loss semaglutide) shows a stronger NAION signal than lower-dose Ozempic (diabetes semaglutide) in FAERS data, greater weight loss, greater blood pressure reduction, greater nocturnal hypotension risk.
Mechanism 3: The “Two-Hit” Model: Drug Effect on a Pre-Vulnerable Nerve
The most widely accepted mechanistic framework is a “two-hit” or multiple-hit model:
- Hit 1 (structural predisposition): A small, crowded optic disc with minimal cup-to-disc ratio, the “disc at risk,” creates a baseline compartment syndrome vulnerability. Any swelling within the optic nerve canal triggers a vicious cycle of edema - compression - ischemia - more edema
- Hit 2 (diabetes/systemic vascular disease): Chronic microvascular damage from diabetes, hypertension, and dyslipidemia reduces the perfusion reserve of the optic nerve head, the margin of safety between normal perfusion and ischemia narrows progressively
- Hit 3 (GLP-1 drug): The acute pharmacological effects of GLP-1 therapy, rapid glucose reduction, blood pressure lowering, plasma volume contraction, and GLP-1 receptor-mediated vascular effects, deliver the final reduction in optic nerve head perfusion that crosses the ischemic threshold in susceptible individuals
In this model, GLP-1 drugs are neither necessary nor sufficient to cause NAION in every patient, but they are a precipitating cause in patients who already have hits 1 and 2. This is a recognized legal concept: a drug does not need to be the sole cause of an injury to be a legally actionable cause.
Mechanism 4: Direct Neurotoxicity: The Rechallenge Evidence
One particularly striking case report cited in the ophthalmology literature involves a patient who developed NAION twice, once after each of two separate semaglutide injections. This rechallenge-dechallenge-rechallenge pattern, the same patient experiencing the same adverse event on repeated exposures to the same drug, is considered among the strongest available evidence of direct drug causation in individual cases. It is difficult to attribute two temporally correlated events to coincidence or confounding.
This single case does not prove population-level causation, but it strongly supports the existence of a subgroup of patients with exceptional optic nerve susceptibility to GLP-1 receptor activation.
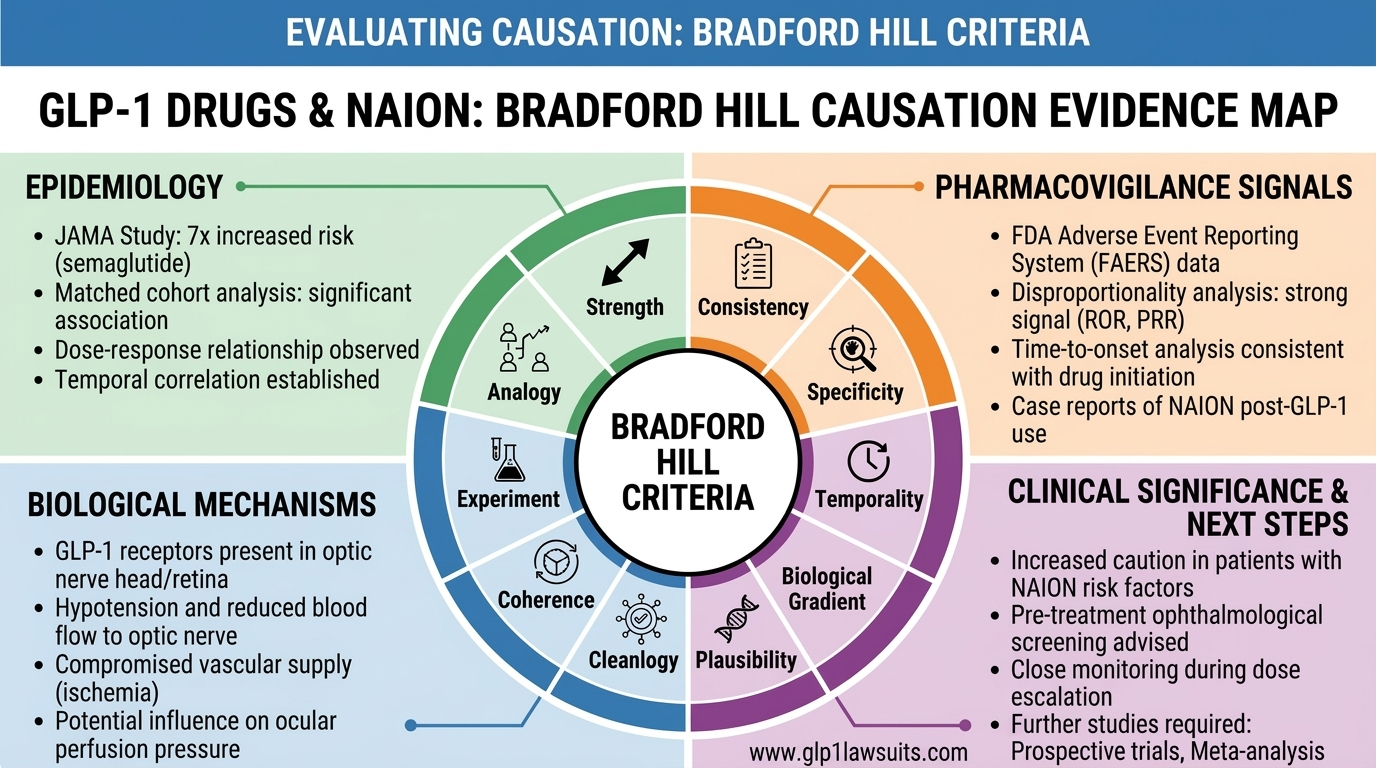
Part IV: The Bradford Hill Causation Analysis
The Bradford Hill criteria, nine viewpoints formulated by Sir Austin Bradford Hill in 1965 to assess whether an epidemiological association is likely to be causal, remain the gold standard framework for causation analysis in pharmaceutical litigation and regulatory science. Here is how the GLP-1/NAION evidence maps onto each criterion.
Important note: Bradford Hill’s criteria are not a checklist where every box must be checked. They are a framework for weighing the totality of evidence. Temporality is the only criterion considered essential, the exposure must precede the outcome. All other criteria strengthen or weaken the causal inference when present.
Criterion 1: Strength of Association
Is the observed association large enough to be unlikely due to chance or confounding?
Evidence: The published studies report hazard ratios and relative risks ranging from 1.34 to 7.64:
- Hathaway (JAMA Ophthalmology): HR 4.28 (diabetes) and 7.64 (obesity)
- Danish nationwide cohort: HR 2.19
- Semaglutide/tirzepatide cohort: HR 1.76
- Meta-analytic pooled: HR 2.78
- FAERS (Wegovy): Reporting odds ratio 74.89
- WHO pharmacovigilance: 68.6× increased reporting rate
Assessment: Strong; associations in the range of 2–8× exceed the threshold typically required for consideration as a strong causal signal in epidemiology. The FAERS and WHO pharmacovigilance signals are extraordinary by any benchmark.
Criterion 2: Consistency
Has the association been observed repeatedly across different populations, settings, and study designs?
Evidence: Positive associations have been observed across:
- Single-center U.S. academic hospital cohorts (Hathaway)
- Nationwide Danish administrative health registry data
- Large U.S. commercial insurance database cohorts
- Global WHO pharmacovigilance data (117,173 patients)
- FDA FAERS spontaneous reporting (30+ million reports)
- Meta-analyses combining multiple study populations
The elevated risk has been observed in both type 2 diabetes and obesity populations, in both semaglutide and tirzepatide users, and in studies using multiple different comparator groups.
Assessment: Consistent; with the noted exceptions of the Sentinel analysis and one other study. The majority of large, well-designed studies find elevated risk.
Criterion 3: Specificity
Is the association specific; does GLP-1 exposure predict NAION specifically, rather than all optic nerve or all vascular events?
Evidence: The semaglutide/tirzepatide cohort study (2025) found that while GLP-1 drugs were associated with increased NAION risk (HR 1.76), they were not associated with increased risk of optic neuritis, papilledema, or other optic nerve conditions. The signal is specific to the ischemic optic neuropathy phenotype, which matches the known vascular mechanism.
Assessment: Moderately specific; the association preferentially targets ischemic optic neuropathy, consistent with a vascular mechanism rather than a general neurotoxic or inflammatory effect.
Criterion 4: Temporality
Did GLP-1 exposure precede NAION onset? (This is the only essential criterion)
Evidence: In every study that includes temporal data:
- The Hathaway study found the risk elevation was most prominent within the first year of semaglutide initiation
- The semaglutide/tirzepatide cohort found that cumulative incidence curves began diverging immediately after medication initiation
- The rechallenge case report documents NAION onset occurring shortly after each of two separate drug exposures
- NAION patients in case reports consistently describe symptom onset occurring after GLP-1 initiation or dose escalation, not before
Assessment: Satisfied; temporal precedence is consistently established. GLP-1 exposure precedes NAION onset in all reported cases.
Criterion 5: Biological Gradient (Dose-Response)
Does increasing the dose or duration of GLP-1 exposure increase NAION risk?
Evidence: This is one of the most significant emerging findings:
- Wegovy (2.4 mg/week semaglutide) ROR 74.89 vs. Ozempic (1.0–2.0 mg/week) ROR 18.81 in FAERS — a 4-fold higher signal at higher doses
- Multivariable adjusted OR for Wegovy vs. Ozempic: 4.74, adjusting for other factors, Wegovy confers nearly 5× the NAION signal of Ozempic
- The 5-year follow-up study found persistent elevated risk with continued GLP-1 use over time
Assessment: Emerging dose-response signal, the Wegovy/Ozempic FAERS differential is the strongest current evidence of a dose-response relationship, and it is consistent with the osmotic/vascular mechanism theories (greater weight loss and blood pressure reduction at higher doses and greater optic nerve perfusion reduction).
Criterion 6: Biological Plausibility
Is there a biologically plausible mechanism by which GLP-1 drugs could cause NAION?
Evidence: Multiple peer-reviewed mechanistic hypotheses have been published:
- Rapid glucose lowering - osmotic optic nerve edema - compartment syndrome
- GLP-1 receptor-mediated vasodilation + blood pressure reduction - nocturnal hypotension - reduced optic nerve perfusion
- Plasma volume contraction from weight loss - reduced effective perfusion pressure
- GLP-1 receptor expression in nervous system tissue including the optic nerve - supporting a direct pharmacological interaction
- Rechallenge/dechallenge case evidence supporting a direct drug effect
Assessment: Plausible; the 2025 Bradford Hill pharmacovigilance analysis (Nature) explicitly concluded that biological plausibility “is supported through GLP-1R expression in the nervous system.” No mechanism has been definitively proven, but multiple converging and biologically coherent hypotheses exist, as is typical at this stage of emerging drug safety science.
Criterion 7: Coherence
Does the causal hypothesis conflict with known facts about NAION or GLP-1 pharmacology?
Evidence: The GLP-1/NAION causal hypothesis is coherent with:
- Known NAION pathophysiology (reduced optic nerve head perfusion in structurally vulnerable patients)
- Known GLP-1 pharmacological effects (vasodilation, blood pressure reduction, rapid glucose lowering)
- Known risk factors for NAION (diabetes, hypertension, nocturnal hypotension, obstructive sleep apnea, all of which are common in GLP-1 users)
- The pattern of NAION (morning onset, unilateral, occurring after initiation of therapy) matching the nocturnal hypotension mechanism
Assessment: Coherent; the GLP-1/NAION causal hypothesis fits within established scientific frameworks.
Criterion 8: Experiment
Is there experimental evidence (controlled trials, animal models, in vitro studies) supporting the association?
Evidence: This is the weakest criterion for the GLP-1/NAION association, as is typical in drug safety litigation:
- No prospective randomized controlled trial has been specifically designed to test GLP-1/NAION causation (an ethics committee would not approve deliberately exposing patients to a potentially blinding drug)
- The rechallenge case provides the closest available evidence to a human experimental confirmation
- Large cardiovascular outcomes trials (SUSTAIN, LEADER, SURPASS) did not specifically track NAION as an endpoint, though some reported ocular adverse events
Assessment: Limited direct experimental evidence; expected at this stage; RCT evidence for drug-specific adverse event causation is rarely available and its absence does not undermine the causal case.
Criterion 9: Analogy
Are there analogous drug/adverse event pairs where a similar mechanism is established?
Evidence: Multiple analogous mechanisms exist:
- Phosphodiesterase type-5 inhibitors (sildenafil/Viagra) cause NAION through a vasodilatory mechanism reducing optic nerve head perfusion - a directly analogous drug/mechanism/outcome triad already established in the pharmacological literature
- Amiodarone-associated optic neuropathy - a medication-induced optic nerve toxicity with partial mechanistic overlap
- Insulin neuritis - rapid glucose correction causing optic nerve edema and ischemia, mechanistically parallel to the rapid glucose lowering hypothesis for GLP-1/NAION
Assessment: Analogous mechanisms established - the sildenafil/NAION analogy is particularly powerful because it is a precedent in which drug-induced vasodilatory effects on optic nerve perfusion are already accepted science and legal precedent.
Bradford Hill Summary Assessment
| Criterion | Evidence | Assessment |
|---|---|---|
| 1. Strength | HR 1.76–7.64; FAERS ROR 74.89; WHO 68.6× | Strong |
| 2. Consistency | 6+ major studies; multiple populations; WHO + FAERS | Consistent (with exceptions) |
| 3. Specificity | NAION-specific signal; no increase in optic neuritis | Moderate |
| 4. Temporality | Drug precedes NAION in all reported cases | Established (essential criterion met) |
| 5. Dose-Response | Wegovy ROR 74.89 vs. Ozempic 18.81; adj. OR 4.74 | Emerging signal |
| 6. Plausibility | Osmotic, vascular, GLP-1R expression mechanisms | Plausible |
| 7. Coherence | Fits NAION pathophysiology and GLP-1 pharmacology | Coherent |
| 8. Experiment | Rechallenge case only; no RCT (ethically impossible) | Limited |
| 9. Analogy | Sildenafil/NAION; insulin neuritis; amiodarone | Strong analogies |
Overall Bradford Hill assessment: Six of nine criteria are satisfied, one shows an emerging signal, one is limited (as expected for any drug adverse event), and the essential criterion (temporality) is fully established. This is a compelling, though not yet conclusive at the population level, causal picture, consistent with where the science stood for many drug-injury associations at comparable stages of litigation and regulatory review.
Part V: What the Regulators Concluded
Regulatory agencies conduct their own causation assessments independently of the academic literature, using both published studies and confidential pharmacovigilance data from manufacturers. Their conclusions carry enormous weight in litigation as evidence of what the defendants knew or should have known.
European Medicines Agency (EMA/PRAC) - June 2025
Following its review, the EMA’s Pharmacovigilance Risk Assessment Committee (PRAC):
- Formally classified NAION as a “very rare” side effect of semaglutide products including Ozempic, Wegovy, and Rybelsus
- Directed that European labels be updated to include NAION as a recognized adverse reaction
- Recommended that semaglutide be discontinued if NAION is confirmed in a patient
- Updated risk management plans for Novo Nordisk’s semaglutide products
World Health Organization (WHO) - June 2025
Following its independent safety review:
- Issued a global safety communication on semaglutide and NAION
- Noted the accumulating evidence from multiple global pharmacovigilance sources
- Recommended healthcare provider and patient awareness of NAION as a potential complication
FDA - Status as of March 2026
As of the date of this article, the FDA has not required a NAION warning on U.S. GLP-1 drug labels. This regulatory gap between European and American label warnings is a central theme in MDL 3163 plaintiffs’ failure-to-warn claims.
The argument: if the EMA and WHO reviewed the same global pharmacovigilance data and concluded the NAION risk was real and required a warning, and Novo Nordisk and Eli Lilly have access to all of this data, then manufacturers could have and should have updated U.S. labels to warn prescribers and patients. Under Wyeth v. Levine, branded drug manufacturers can unilaterally update their labels using the FDA’s “Changes Being Effected” (CBE) regulation without prior FDA approval. The absence of a U.S. warning is a choice, not a regulatory prohibition.
Part VI: The Manufacturer Knowledge Timeline; What Novo Nordisk and Eli Lilly Knew and When
One of the most critical issues in MDL 3163 is the manufacturer knowledge timeline: when did Novo Nordisk and Eli Lilly know, or should have known, about the NAION risk? The discovery process in the MDL is expected to expose internal documents, pharmacovigilance reports, medical affairs communications, labeling discussions, that reveal the answer.
What is publicly known already:
| Date | Event |
|---|---|
| 2012 | At least one plaintiff’s complaint alleges that reports of optic ischemic neuropathy associated with GLP-1 drugs existed as far back as this date - well before the JAMA studies |
| 2017–2024 | FDA FAERS accumulates adverse event reports linking semaglutide to ischemic optic neuropathy - data that Novo Nordisk receives and is required to analyze as part of its pharmacovigilance obligations |
| July 2024 | Hathaway JAMA Ophthalmology study published - the first high-profile peer-reviewed study; the general causation debate becomes public |
| June 2025 | EMA/WHO formally recognize NAION as semaglutide side effect; European labels updated |
| As of March 2026 | No FDA-required NAION warning on U.S. labels |
The gap between what manufacturers knew from their own pharmacovigilance data and what was disclosed to U.S. prescribers and patients will be one of the most heavily contested issues at trial.
What This Causation Analysis Means for Your Legal Claim
General Causation Is the Foundation
A plaintiff in MDL 3163 first needs to establish general causation, that GLP-1 drugs as a class can cause NAION in some patients. The evidence reviewed above, eight major studies, regulatory recognition from EMA and WHO, strong Bradford Hill criterion satisfaction, and multiple biologically plausible mechanisms, supports a general causation opinion from qualified ophthalmology and epidemiology experts.
The FDA Sentinel null result and other conflicting studies create a Daubert battleground, but courts routinely accept expert general causation opinions where the weight of evidence supports the association, even when the scientific debate is not fully resolved.
Specific Causation Connects the Evidence to Your Case
Beyond general causation, each plaintiff must establish specific causation, that this patient’s NAION was caused by this drug. Key elements of specific causation include:
- Temporal relationship: NAION onset occurring after GLP-1 initiation or dose escalation
- Differential diagnosis: Ruling out other independent causes through comprehensive workup
- Risk factor overlap: Documenting the specific “hits” (disc at risk, diabetes, nocturnal hypotension) that the GLP-1 drug’s effects pushed across the ischemic threshold
- No alternative explanation: The absence of other events or exposures that independently explain the NAION
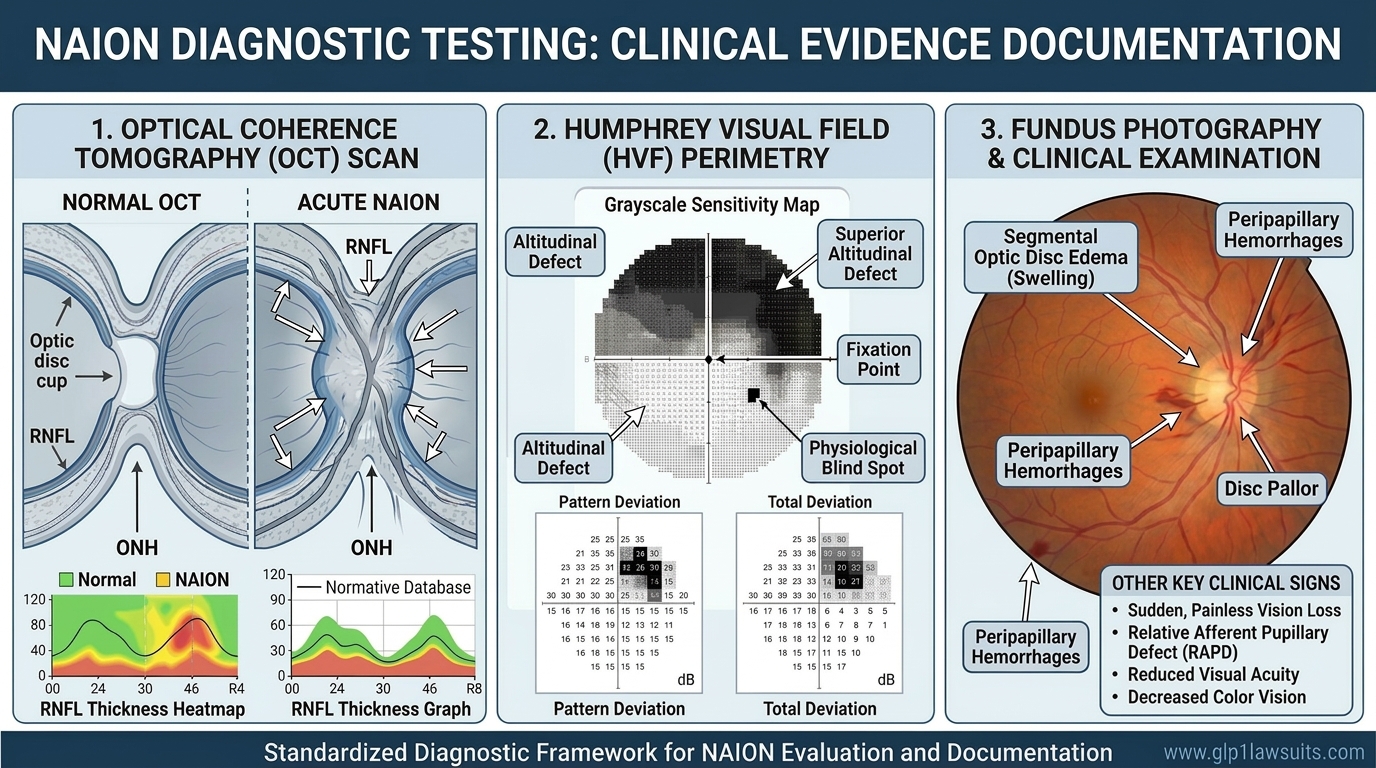
For the diagnostic tests that document specific causation, read: NAION Eye Tests Required for Vision Loss Claims
The Regulatory Gap as Failure-to-Warn Evidence
Perhaps the most litigation-ready piece of this causation analysis is the regulatory asymmetry: the EMA recognized NAION as a semaglutide adverse effect and required a label update in June 2025; the FDA has not followed. This gap is powerful evidence that:
- The scientific and pharmacovigilance data was sufficient to support a warning
- The manufacturer had notice of the risk through its own global pharmacovigilance program
- U.S. prescribers and patients were denied information that European prescribers and patients received
For the full litigation update, read: NAION MDL Update: GLP-1 Vision Loss Litigation Status
Q&A: GLP-1 Drugs, Diabetes, and NAION Causation
Q1. Does the science prove that GLP-1 drugs cause NAION?
The science shows a consistent, replicated, statistically significant association between GLP-1 drugs and increased NAION risk across multiple studies and populations, supported by biologically plausible mechanisms, formal regulatory recognition from the EMA and WHO, and extraordinary pharmacovigilance signals. “Proof” in the sense of a definitive randomized controlled trial does not exist and is ethically impossible to obtain. The current evidence meets the Bradford Hill causation framework to a degree sufficient to support expert causation opinions in litigation, consistent with the evidentiary standard applied in most pharmaceutical MDLs.
Q2. If diabetes already causes NAION, how can GLP-1 drugs be blamed?
Diabetes is an independent NAION risk factor, but that does not mean GLP-1 drugs cannot be an additive cause. In tort law, a drug does not need to be the sole cause of an injury; it needs to be a contributing cause that materially increased the risk. The epidemiological evidence, particularly studies using other diabetic patients on different medications as comparators, shows that GLP-1 users develop NAION at significantly higher rates than similarly diabetic patients on other drugs. The increased risk is attributable to the GLP-1 drug, not merely the underlying diabetes.
Q3. Why does Wegovy seem to have a stronger NAION signal than Ozempic?
Both drugs contain semaglutide, but Wegovy is prescribed at higher doses (up to 2.4 mg/week vs. 1.0–2.0 mg/week for Ozempic) and is used primarily for weight loss rather than diabetes management, meaning Wegovy patients often experience more rapid and pronounced weight loss, greater blood pressure reduction, and potentially more significant plasma volume contraction. All of these effects may amplify the reduction in optic nerve head perfusion. The FAERS data showing a nearly 5× higher adjusted NAION risk for Wegovy vs. Ozempic is consistent with a dose/effect-magnitude relationship.
Q4. Does the FDA’s failure to require a NAION warning mean GLP-1 drugs are safe?
Not necessarily. Regulatory approval and label adequacy are separate questions. The FDA’s inaction on a NAION warning as of early 2026 contrasts with the EMA and WHO, which both acted in June 2025. Under Wyeth v. Levine, branded drug manufacturers can and must update their labels when new safety information warrants it, without waiting for FDA direction. The EMA and WHO conclusions, combined with the published literature and manufacturers’ own pharmacovigilance obligations, support the argument that Novo Nordisk and Eli Lilly could and should have acted sooner.
Q5. What is the absolute risk of NAION from GLP-1 drugs?
The 5-year retrospective cohort study found an absolute 5-year NAION risk of 0.087% in GLP-1 users, roughly 1 in 1,150 patients over five years. The semaglutide/tirzepatide cohort found 0.04% NAION incidence at 2 years vs. 0.02% in the comparison group. These absolute numbers are small relative to GLP-1’s benefits for most patients, but they represent thousands of patients when applied to the tens of millions of GLP-1 prescriptions filled annually in the U.S., and each event carries catastrophic personal consequences because NAION is largely irreversible.
Q6. Does the conflicting Sentinel study mean my case has no merit?
Conflicting studies are a normal feature of emerging drug safety science, not an automatic barrier to recovery. Courts and juries evaluate the totality of evidence, including the weight of multiple studies, the regulatory response of the EMA and WHO, the pharmacovigilance signals, and the biological plausibility of the mechanism. The existence of one or two null studies alongside six or more positive studies is consistent with how pharmaceutical causation battles have been litigated and won in Daubert proceedings across many MDLs. Only a licensed attorney can assess the specific implications for your individual claim.
Why Work With Us
We Understand the Causation Science, and So Will Our Experts
Winning a GLP-1 NAION case requires more than a compelling story, it requires causation experts who can withstand Daubert challenge on the Bradford Hill framework, the epidemiological literature, and the biological mechanisms. We retain and prepare experts who understand both the medicine and the legal standards for scientific admissibility.
Exclusive Focus on GLP-1 Injuries
This is what we do. Our deep familiarity with the evolving science, including the FAERS dose-response signal, the EMA/FDA regulatory gap, and the manufacturer knowledge timeline, means we are positioned to build the strongest possible causation narrative for your claim.
No Upfront Costs
Pure contingency-fee representation: no fees unless we win, all litigation costs advanced, free initial consultation.
Contact Us for a Free Consultation
The science is building. The litigation is open. Your window to file is limited.
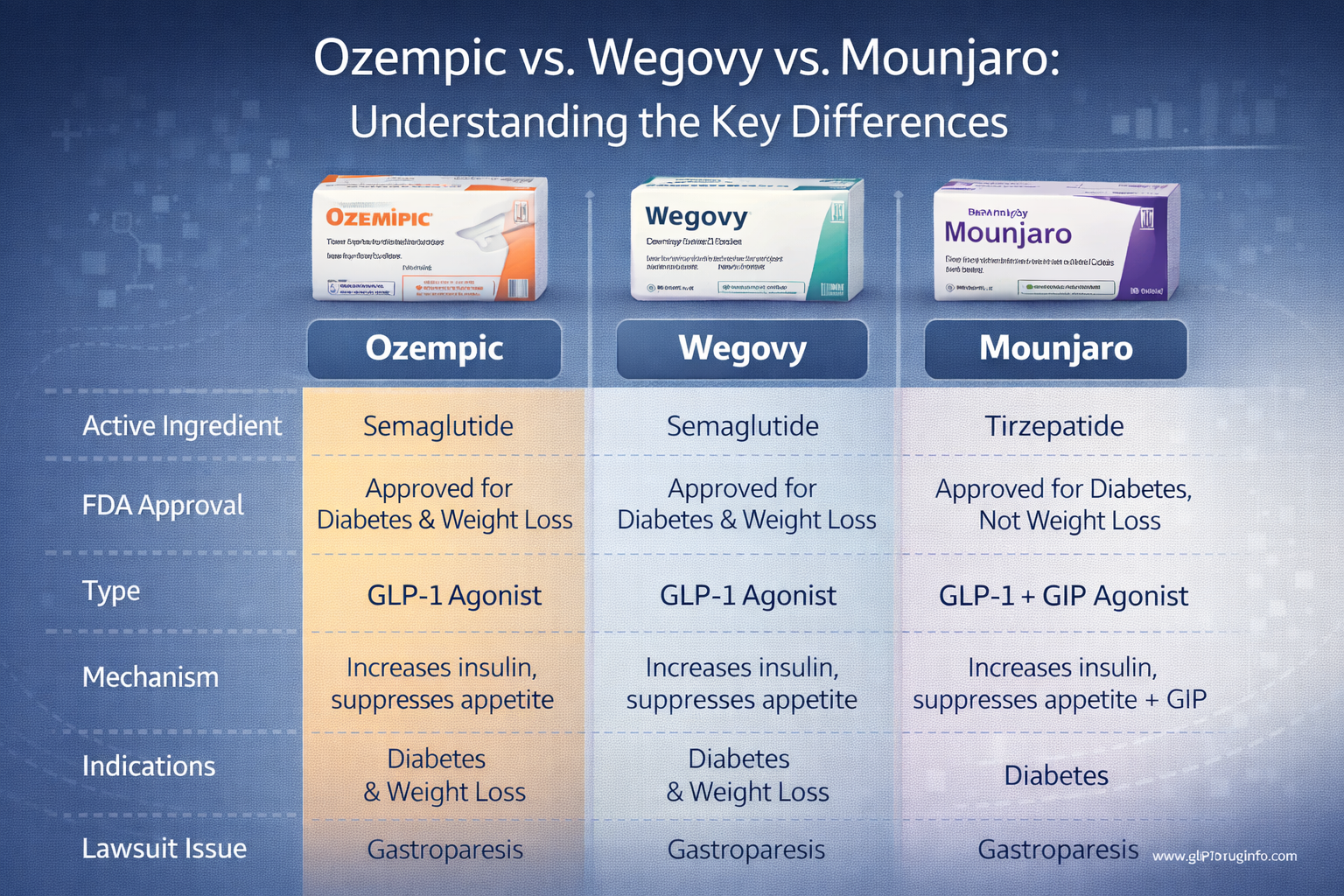
If you took Ozempic, Wegovy, Mounjaro, or Zepbound and developed sudden vision changes or were diagnosed with NAION, MDL 3163 is open and accepting new cases. The statute of limitations is running.
Visit www.GLP1Lawsuits.com to:
- Get a free case evaluation
- Speak with an experienced pharmaceutical-litigation attorney about your NAION case
- Learn more about MDL 3163 and causation evidence in your claim
- Get a timeline-specific analysis of your GLP-1 use, diagnosis, and injury
- Join others seeking accountability for preventable, permanent vision loss
No upfront fees. No costs unless we win your case. Don’t let the statute of limitations run out.
Sources Cited
| # | Authority | Key Data |
|---|---|---|
| 1 | Hathaway et al. - JAMA Ophthalmology (July 2024) | HR 4.28 (diabetes), 7.64 (obesity); temporal risk within first year; 16,827 patients; first major peer-reviewed study |
| 2 | Danish Nationwide Cohort (2024) | 424,152 patients; HR 2.19 (95% CI 1.54–3.12); nationwide administrative data |
| 3 | Semaglutide/Tirzepatide Cohort - PMC/JAMA Network Open (2025) | 159,398 matched patients; HR 1.76; NAION 0.04% vs. 0.02%; cumulative incidence diverges at day one; extends risk to tirzepatide |
| 4 | Five-Year Retrospective Cohort - PMC (2026) | 776,666 patients; RR 1.339 (p=0.005); 5-year absolute risk 0.087%; E-value sensitivity analysis confirms robustness |
| 5 | GLP-1RA vs. SGLT2 Inhibitors + Meta-Analysis (2026) | HR 1.85 (new study); meta-analytic pooled HR 2.78 (95% CI 1.39–5.56); consistent across subgroups |
| 6 | FDA FAERS Pharmacovigilance - Nature (2025) | 30+ million FAERS reports; Wegovy ROR 74.89; Ozempic ROR 18.81; men ROR 116.37; adj. OR Wegovy vs. Ozempic 4.74; sex-stratified findings |
| 7 | WHO Global Pharmacovigilance - AAO 2025 | 117,173 patients; semaglutide 68.6× more likely NAION vs. comparators; global real-world signal |
| 8 | Bradford Hill Pharmacovigilance Analysis - Nature (2025) | Explicit Bradford Hill application: biological plausibility via GLP-1R expression; temporal consistency confirmed |
| 9 | FDA Sentinel Analysis (September 2025) | Large network study; no statistically significant increased risk; key defense evidence; comparator and ascertainment methodology differences cited |
| 10 | PMC - Five-Year Fellow Eye Study (2025) | Diabetes shortens fellow-eye attack interval from 122 months to 14 months - 8× compression; dyslipidemia OR 8.36 |
| 11 | PMC - NAION Risk Factors in T2DM (2025) | Dyslipidemia OR 8.36; LDL OR 1.017; small cup-to-disc ratio OR 11.92 as independent NAION risk factors in diabetes |
| 12 | Cureus / PMC - Osmotic Mechanism Review (2026) | Rapid glucose lowering - intravascular osmotic pressure drop - optic nerve edema mechanism; comparison to insulin neuritis |
| 13 | Review of Optometry - Rechallenge Case (2025) | Patient developed NAION twice on two separate semaglutide injections - rechallenge-dechallenge-rechallenge evidence |
| 14 | Vision Monday - Expert Commentary (March 2026) | “Microvascular perfusion and optic nerve head vulnerability”; vascular dysregulation hypothesis; nocturnal hypotension mechanism |
| 15 | EMA/PRAC - Safety Communication (June 2025) | Formal NAION classification as very rare semaglutide adverse effect; label update directive; discontinuation recommendation |
| 16 | WHO - Global Safety Communication (June 2025) | Semaglutide and NAION safety alert; healthcare provider and patient awareness recommendation |
| 17 | U.S. Supreme Court - Wyeth v. Levine (2009) | Branded drug manufacturers may unilaterally update labels using CBE regulation; failure-to-warn claims not federally preempted |
| 18 | Bradford Hill Criteria Framework - PMC (2020/2015) | Nine viewpoints for causation assessment; temporality as essential criterion; totality of evidence standard |
Disclaimer
This article provides general scientific and legal information about the causal relationship between GLP-1 drugs and NAION and does not constitute medical or legal advice. The science in this area is evolving; new studies and regulatory decisions may update the picture described here. For evaluation of your medical situation, consult a qualified ophthalmologist, neuro-ophthalmologist, or other healthcare provider. For evaluation of your legal rights, consult a licensed pharmaceutical litigation attorney who can assess your specific facts, records, and applicable deadlines. Each case is unique; past results do not guarantee future outcomes.
© 2026 GLP1lawsuits.com | All Rights Reserved
Last reviewed by a licensed attorney: March 2026