NAION is often called an “optic-nerve stroke.” It strikes without warning, usually overnight, and the vision it takes rarely comes back.
Non-arteritic anterior ischemic optic neuropathy (NAION) is the most common cause of acute, permanent optic-nerve damage in adults over 50, and it is now at the center of a growing wave of lawsuits against the makers of GLP-1 drugs such as Ozempic, Wegovy, Mounjaro, and Zepbound. This article explains exactly what NAION is, how it damages the optic nerve, who is most vulnerable, and why the emerging link to GLP-1 medications matters for patients and their families.
📖 For context on why GLP-1 drugs are being linked to this condition, read our companion article: Understanding the Growing Concerns Around GLP-1 Drugs and Vision Loss (NAION)
What Does NAION Stand For?
The full name, non-arteritic anterior ischemic optic neuropathy, describes exactly what the condition is:
| Term | Meaning |
|---|---|
| Non-arteritic | Not caused by giant cell arteritis (GCA), a systemic inflammatory disease |
| Anterior | Affects the front (anterior) portion of the optic nerve, just behind the eye |
| Ischemic | Caused by a sudden loss of adequate blood flow (ischemia), like a stroke |
| Optic neuropathy | Damage to the optic nerve, the cable that carries visual signals from the eye to the brain |
Put simply, NAION is a vascular event in the optic nerve, an interruption in blood supply that destroys nerve fibers and produces sudden, often permanent vision loss.
NAION vs. AAION: A Critical Distinction
There are two main types of anterior ischemic optic neuropathy. Distinguishing between them is medically urgent because one requires emergency treatment for a life-threatening systemic disease.
| Feature | NAION (Non-Arteritic) | AAION (Arteritic) |
|---|---|---|
| Cause | Vascular/structural factors; now linked to GLP-1 drugs | Giant cell arteritis (GCA), a systemic vasculitis |
| Age group | Typically 50–70 | Typically 70+ |
| Pain | Painless | Often jaw pain, scalp tenderness, headache |
| Vision loss severity | Moderate to severe | Often profound (“no light perception”) |
| Disc appearance | Hyperemic (pink/red), swollen | Pale, chalky white swelling |
| Lab markers | Normal ESR/CRP | Markedly elevated ESR/CRP |
| Treatment | No proven reversal; risk-factor management | Urgent systemic corticosteroids to preserve the other eye |
| Emergency level | Urgent evaluation needed | True medical emergency |
Because the two conditions can look similar on exam, any patient with sudden vision loss and a swollen optic disc must receive immediate ophthalmologic evaluation and blood tests to rule out AAION before assuming NAION.
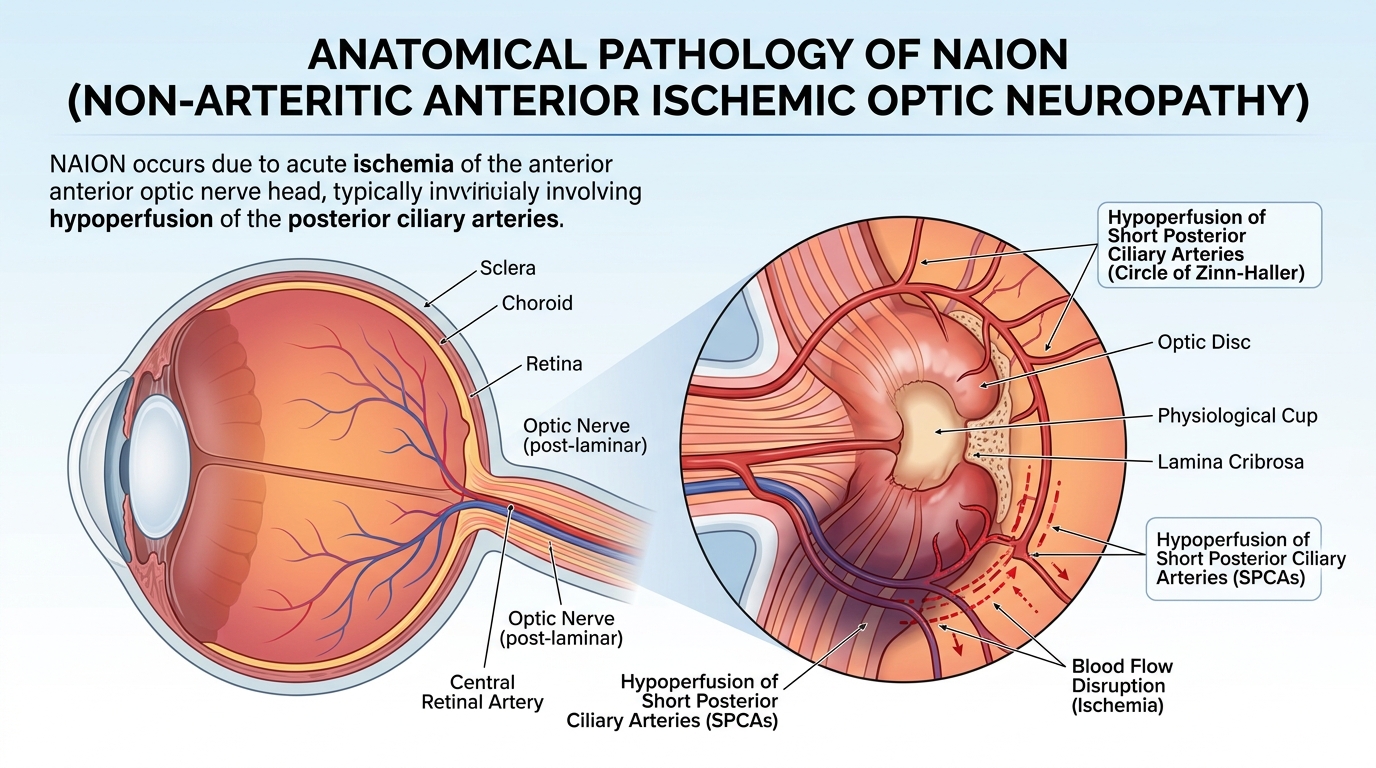
How NAION Damages the Optic Nerve: The Pathophysiology
Understanding why NAION occurs helps explain why GLP-1 users, many of whom already have diabetes, hypertension, or sleep apnea, may be at elevated risk.
Step 1: The Optic Nerve’s Vulnerable Blood Supply
The front portion of the optic nerve is fed almost entirely by small vessels called the short posterior ciliary arteries, which branch from the ophthalmic artery. These vessels form an arterial ring (the Circle of Zinn-Haller) around the optic nerve as it passes through a narrow opening in the back of the eye. Unlike most brain tissue, this region has minimal backup collateral blood supply, making it uniquely vulnerable to any drop in perfusion pressure.
Step 2: The “Disc-at-Risk” Anatomy
In people with a small, crowded optic disc, called a “disc-at-risk” or a low cup-to-disc ratio of 0.2 or less—the optic nerve fibers are tightly packed within a rigid scleral canal. This anatomical variant is present in over 90% of patients who develop NAION and is often bilateral, meaning the fellow eye carries the same structural risk. When ischemia causes even minor swelling inside this confined space, the result is a compartment-syndrome-like cascade: swelling compresses neighboring axons, which cuts off their blood supply further, which causes more swelling, a vicious cycle that ends in irreversible nerve-fiber death.
Step 3: Nocturnal Hypotension and the “Wake-Up Stroke”
Approximately 70–80% of NAION episodes are first noticed upon awakening, strongly pointing to nighttime low blood pressure (nocturnal hypotension) as a key trigger. During sleep, blood pressure normally dips. In susceptible patients, especially those taking antihypertensive medications at bedtime, this drop may push perfusion pressure in the already-vulnerable optic nerve below a critical threshold. The result is localized optic-nerve infarction that the patient discovers when they open their eyes in the morning.
Step 4: Axoplasmic Stasis, Edema, and Nerve-Fiber Death
Once blood flow is compromised, the flow of nutrients and cellular cargo along nerve-fiber axons (axoplasmic flow) is disrupted. Axons swell with accumulated organelles and waste products. Inside the rigid scleral canal, this swelling cannot be accommodated, compressing adjacent axons and their microvasculature. Ganglion cells, whose axons make up the optic nerve, then undergo apoptosis (programmed cell death). Over weeks to months, the swollen disc resolves into pallor, and the retinal nerve fiber layer visibly thins on imaging, a sign of permanent structural loss.
How GLP-1 Drugs May Fit Into This Mechanism
Researchers and plaintiff experts in GLP-1 litigation have proposed several ways in which semaglutide and tirzepatide may interact with this vulnerability:
- Rapid blood-pressure reduction: GLP-1 drugs lower blood pressure; aggressive lowering, especially at night, may precipitate nocturnal hypotension in susceptible patients.
- Rapid glycemic improvement: Sudden drops in blood sugar can affect vascular tone and ocular perfusion.
- Vascular effects: GLP-1 receptors are expressed in vascular smooth muscle and endothelial cells; GLP-1 agonism may alter autoregulation of optic nerve blood flow.
- Rapid weight loss: Large, fast reductions in body weight, common with these drugs, may produce hemodynamic shifts affecting small-vessel perfusion.
📖 For a detailed causation analysis, see: The Connection Between Diabetes, GLP-1 Drugs and NAION
Who Gets NAION? Epidemiology and Statistics
NAION is more common than most patients and many clinicians realize.
- Annual incidence in the United States: approximately 2.3 to 10.3 cases per 100,000 individuals aged 50 or older
- Most common acute optic neuropathy in adults over 50, more frequent than the arteritic form (AAION), which occurs at about 0.36 per 100,000 per year
- Peak age range: 60–70 years, though cases in younger adults are documented, especially when sleep apnea, migraines, or clotting disorders are present
- Sex: Slight male predominance in most studies, possibly due to higher rates of cardiovascular disease and sleep apnea
- Race: More common in Caucasian populations, likely because of a higher prevalence of the disc-at-risk anatomy; less common in African American individuals, who statistically have larger cup-to-disc ratios
- Bilateral risk: Although NAION is unilateral at onset, the second eye is affected in 15–25% of patients within five years, especially when systemic risk factors remain uncontrolled
These figures were established before GLP-1 drug use became widespread. With tens of millions of patients now taking these medications, researchers and regulators have flagged a potential increase in NAION incidence that may not yet be fully reflected in population statistics.
Risk Factors: Who Is Most Vulnerable?
NAION is a multifactorial disease, meaning multiple conditions must usually align for it to occur. The leading risk factors, as established in peer-reviewed ophthalmology and neurology literature, include:
Structural (Anatomical) Risk Factors
- Disc-at-risk (small cup-to-disc ratio ≤ 0.2): present in over 90% of NAION cases; this is the single strongest individual risk factor
- Optic disc drusen: calcium deposits within the optic nerve head that further crowd the confined canal space
- Shallow anterior chamber or high hyperopia (farsightedness): associated with crowded optic discs
Vascular and Systemic Risk Factors
- Systemic hypertension: damages the microvasculature supplying the optic nerve head and impairs autoregulation
- Diabetes mellitus: causes endothelial dysfunction, basement membrane thickening, and microvascular disease that reduces optic nerve perfusion, the same patients most commonly prescribed GLP-1 medications
- Hyperlipidemia/high cholesterol: contributes to atherosclerosis and vascular stiffness in the posterior ciliary arteries
- Obstructive sleep apnea (OSA): causes repeated nocturnal oxygen drops and blood-pressure swings that stress optic nerve perfusion; strongly and independently associated with NAION
- Atherosclerotic cardiovascular disease: a marker of generalized small-vessel disease
- Nocturnal hypotension: particularly pronounced in patients taking antihypertensives at bedtime
Medication-Related Risk Factors
- Phosphodiesterase-5 (PDE-5) inhibitors such as sildenafil (Viagra) and tadalafil: well-established NAION triggers in susceptible individuals
- GLP-1 receptor agonists including semaglutide and tirzepatide: now under active investigation as potential triggers, with multiple large studies showing a significantly elevated relative risk
Rare and Prothrombotic Risk Factors
In younger patients without classical vascular risks, NAION may be associated with:
- Antiphospholipid antibody syndrome
- Protein C or S deficiency
- Hyperhomocysteinemia
- Elevated blood viscosity (polycythemia vera, Waldenstrom macroglobulinemia)
Symptoms of NAION: What Patients Experience
NAION has a distinctive clinical presentation that sets it apart from many other causes of vision loss.
Classic Onset
- Sudden, painless vision loss in one eye, typically discovered upon awakening in the morning
- Vision loss may be noticed all at once or worsen over hours to a few days before stabilizing
- No associated eye pain, redness, or headache (unlike optic neuritis, which is painful)
Visual Acuity
- Visual acuity at presentation ranges widely, from near-normal (20/20) to severe (20/200 or worse), but most patients fall in the 20/60 to 20/200 range
- About half of patients retain relatively good central sharpness initially, even with significant field loss
Visual Field Defects
- The most characteristic pattern is an inferior altitudinal defect: loss of the lower half of the visual field in the affected eye
- Superior altitudinal, arcuate, and diffuse field losses also occur
- Central and paracentral scotomas (blind spots) may be present
Other Symptoms and Signs
- Loss or dulling of color vision (dyschromatopsia) in the affected eye
- Relative afferent pupillary defect (RAPD): when a light is shone in the affected eye, both pupils constrict less briskly, a reliable clinical sign of optic-nerve damage
- On fundus exam in the acute phase: sectoral or diffuse optic disc swelling, often with peripapillary flame-shaped hemorrhages and absence of disc cupping (unlike glaucoma)
- Over weeks to months, the swelling resolves and is replaced by optic disc pallor (pale appearance), indicating permanent nerve fiber loss
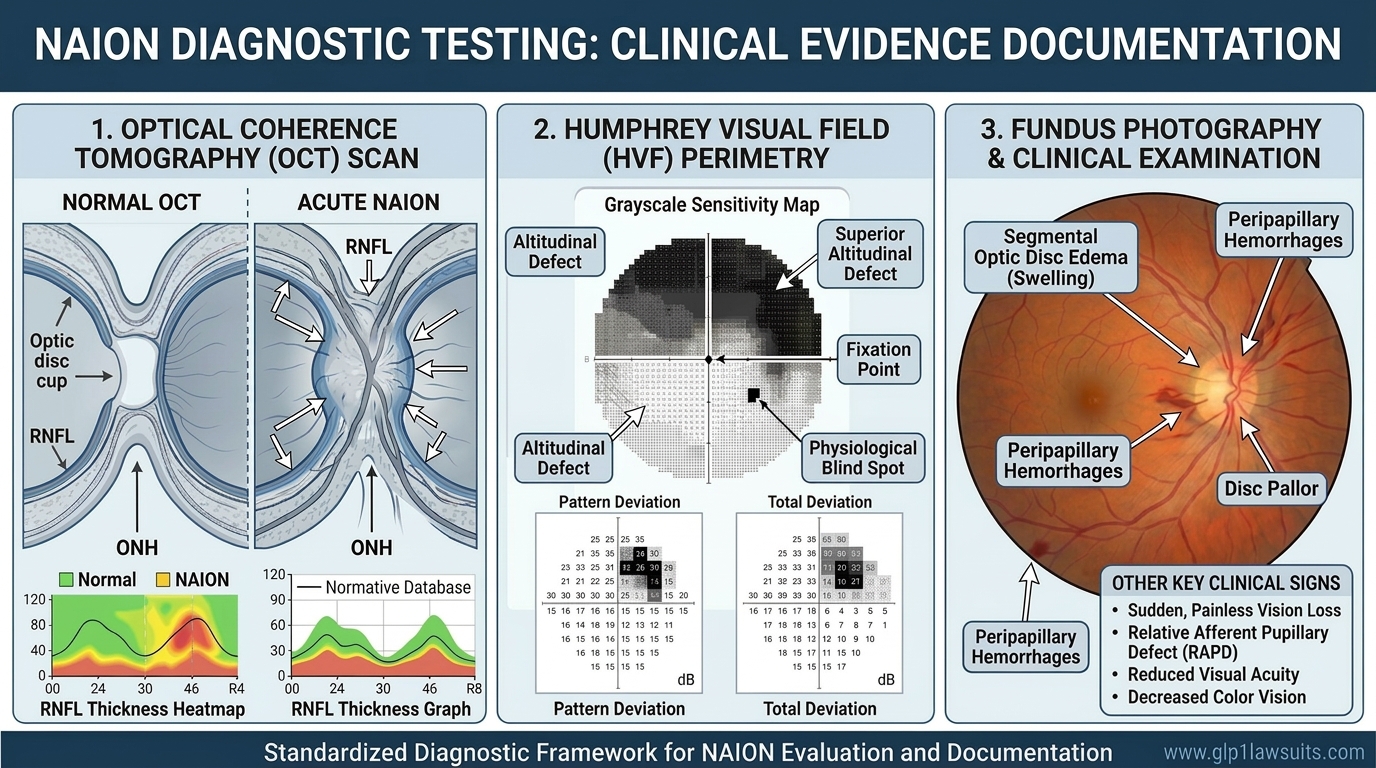
How Is NAION Diagnosed?
NAION is primarily a clinical diagnosis based on the history and a detailed ophthalmic examination. There is no single definitive test; rather, the diagnosis involves ruling out other causes and documenting the characteristic findings.
Essential Components of the NAION Workup
- Detailed history: Sudden vision loss, time of onset (morning?), known risk factors, current medications including GLP-1 drugs
- Visual acuity testing: Baseline documentation of how much central vision remains
- Visual field testing (perimetry): Maps the pattern and severity of field loss; inferior altitudinal defect is the hallmark
- Dilated fundus examination: The eye doctor examines the optic disc for swelling, hemorrhages, pallor, and cup-to-disc ratio
- Pupillary reflex testing: RAPD is almost always present and is an objective sign of optic nerve dysfunction
- Optical coherence tomography (OCT): High-resolution imaging that documents optic disc edema in the acute phase and retinal nerve fiber layer (RNFL) thinning in the chronic phase—crucial for legal documentation
- Fluorescein angiography (FA): Can show delayed filling of the optic disc in NAION; also helps exclude other conditions such as central retinal vein occlusion
- Laboratory tests: ESR, CRP, and other markers to rule out giant cell arteritis (the arteritic form, which requires emergency treatment)
- MRI of the brain and orbits: Ordered when the diagnosis is uncertain, to exclude demyelinating disease (optic neuritis), compressive lesions, or stroke
Why Documentation Matters for Legal Claims
For patients pursuing a GLP-1 NAION lawsuit, timely and thorough diagnostic documentation is essential. OCT images, visual field printouts, fundus photographs, and pharmacy records linking GLP-1 use to the timeline of vision loss form the medical foundation of a viable legal claim.
📖 For a detailed walkthrough of the tests involved and their legal significance, read: NAION Diagnosis: What Eye Tests Are Involved? and Diagnostic Tests for NAION and Vision Loss: What to Expect
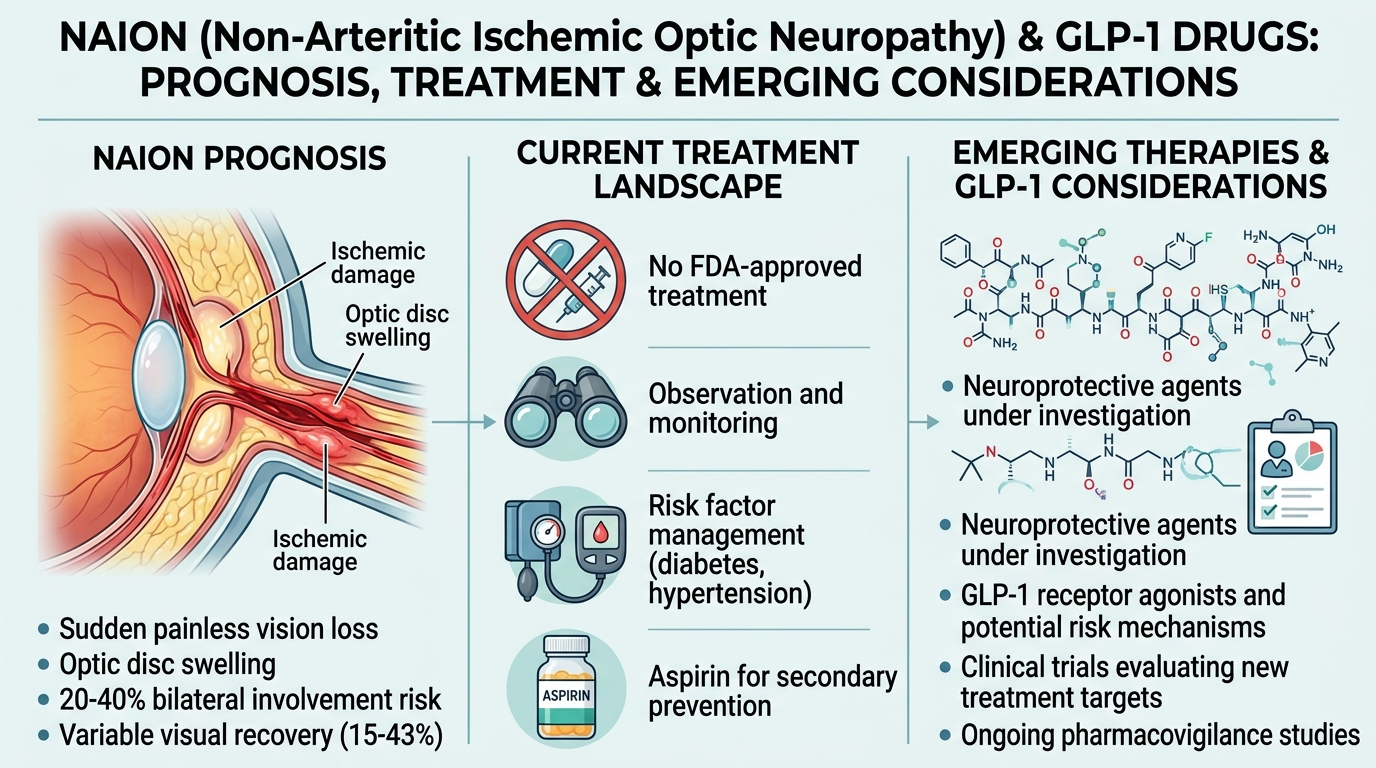
Natural History and Prognosis: What Happens Over Time?
The Ischemic Optic Neuropathy Decompression Trial (IONDT), the largest randomized controlled study of NAION’s natural history, established the following outcomes at two-year follow-up:
- ~30% of patients spontaneously recover 3 or more lines of visual acuity
- ~20% of patients lose an additional 3 or more lines
- ~50% of patients remain stable with no significant change
These figures, while offering some hope for partial recovery, underscore a sobering reality: the majority of patients do not regain meaningful vision, and some continue to deteriorate. Visual field defects, which are often more functionally disabling than reduced acuity, generally do not improve significantly even when acuity stabilizes.
Apparent improvements in visual acuity over time may partly reflect fixation adaptation, the brain learning to use adjacent healthy retinal areas, rather than true nerve regeneration.
For patients whose fellow eye is also a disc-at-risk, the cumulative risk of bilateral NAION within 5 years is 15–25%, especially when systemic risk factors remain uncontrolled.
📖 For a full discussion of recovery expectations and treatment options, read: Can NAION Be Reversed? Vision Loss Prognosis and Treatment and Can You Recover From NAION? Realistic Expectations & Prognosis
Current Treatment Landscape: Why There Is No Proven Cure
Despite decades of research, no treatment has been proven in large, well-controlled trials to reliably reverse NAION or restore lost vision. Approaches that have been studied without conclusive benefit include:
- Systemic corticosteroids: Some small studies suggest modest short-term acuity benefit, but data are inconsistent and steroids carry significant risks in diabetic patients (worsened blood sugar) and patients with hypertension
- Anti-VEGF agents (e.g., bevacizumab): Investigated to reduce optic disc swelling; no clear vision benefit demonstrated in randomized trials
- Optic nerve sheath decompression surgery: The IONDT trial showed this procedure was no better than observation and carried a risk of harm; it is no longer recommended
- Hyperbaric oxygen: Anecdotal reports only; not established as effective
- Neuroprotective agents: Under active investigation in research settings; no approved treatment currently available
Current management therefore focuses on:
- Discontinuing suspected triggers (including, per EMA and WHO guidance, stopping semaglutide if NAION is confirmed)
- Optimizing systemic risk factors: blood pressure, blood sugar, cholesterol, and treatment of sleep apnea
- Adjusting antihypertensive timing: If nocturnal hypotension is suspected, moving evening doses to morning may help protect the fellow eye
- Monitoring the fellow eye closely, given the 15–25% risk of bilateral involvement
- Low vision rehabilitation: Working with low vision specialists and occupational therapists to maximize functional independence
NAION, GLP-1 Drugs, and Your Legal Rights
Patients who developed NAION while taking Ozempic, Wegovy, Mounjaro, Zepbound, or other GLP-1 medications may have a legal claim based on allegations that:
- Manufacturers such as Novo Nordisk and Eli Lilly failed to adequately warn prescribers and patients about the risk of NAION
- The drugs were unreasonably dangerous given the known risk of permanent vision loss that was not disclosed on the label
- Plaintiffs suffered permanent, life-altering harm including vision loss, reduced ability to drive and work, and significant reduction in quality of life
A federal MDL has been established to coordinate GLP-1 NAION vision-loss claims, and state-court consolidations are also underway. Strict filing deadlines (statutes of limitation) apply—missing these deadlines can permanently bar an otherwise valid claim.
📖 For the latest litigation developments, see: GLP-1 NAION MDL3163 Update
Q&A What Is NAION and How Does It Relate to GLP-1 Drugs?
Q1. Is NAION the same as a stroke?
NAION is often described as an “optic-nerve stroke” because it results from an interruption of blood flow to the optic nerve, similar to how a cerebral stroke results from interrupted blood flow to the brain. However, NAION is localized to the optic nerve and does not involve the brain directly; it does not cause the limb weakness, speech problems, or other neurological signs of a cerebral stroke.
Q2. How does NAION differ from glaucoma?
Both NAION and glaucoma can damage the optic nerve and cause permanent vision loss, but they are distinct conditions. Glaucoma is typically a chronic, slowly progressive condition associated with elevated intraocular pressure and gradual nerve-fiber loss. NAION is an acute vascular event causing sudden vision loss; the optic disc in NAION typically shows swelling without the characteristic cupping (excavation) seen in glaucoma.
Q3. Can NAION affect both eyes?
Yes. Although NAION always begins in one eye, the fellow eye is affected in approximately 15–25% of cases within five years, particularly when the fellow eye also has a disc-at-risk anatomy and when systemic risk factors such as hypertension, diabetes, and sleep apnea remain uncontrolled.
Q4. Is NAION always permanent?
Not always, approximately 30% of patients experience some spontaneous visual improvement within two years, according to the IONDT. However, significant recovery is the exception rather than the rule, and visual field defects typically persist even when acuity partially improves. Most patients should plan for the likelihood that the vision lost will not fully return.
Q5. I take Ozempic for diabetes. Does that mean I will get NAION?
No. The vast majority of GLP-1 users will not develop NAION. Current estimates suggest NAION may affect up to approximately 1 in 10,000 semaglutide users, a very low absolute risk, though a meaningfully higher relative risk compared to some other diabetes medications. The key is knowing the warning signs, understanding your personal risk factors, and seeking immediate evaluation if sudden vision changes occur.
Q6. What should I do if I think I had NAION while on a GLP-1 drug?
Seek evaluation by an ophthalmologist or neuro-ophthalmologist as soon as possible if you have not already done so, thorough documentation is important both medically and legally. Inform your prescriber of the vision event and discuss whether your GLP-1 medication should be stopped. Preserve all medical records, imaging results, and pharmacy documentation. Then consult a pharmaceutical litigation attorney to understand your rights and the applicable filing deadlines.
Why Work With Us
Exclusive Focus on GLP‑1 Injuries
Our practice is dedicated entirely to GLP‑1 medication injuries, including NAION vision loss, gastroparesis, ileus, and related complications. This is not a side project or add‑on; it is the core of what we do every day.
- Deep, case‑specific knowledge of Ozempic, Wegovy, Mounjaro, Zepbound, and related medications
- Familiarity with how these drugs work, their known side‑effect profiles, and the evolving medical literature around NAION and other serious harms
- Hands‑on experience with MDL No. 3094 and MDL 3163 procedures, case‑management orders, and plaintiff requirements in complex pharmaceutical litigation
Because we live in this space, we can quickly identify the strengths and weaknesses of a potential GLP‑1 claim and tailor your case strategy around the most up‑to‑date science and MDL developments.
Integrated Medical Expert Network
GLP‑1 and NAION cases are built on medicine as much as law. We work closely with a network of highly qualified specialists who understand these specific injuries:
- Board‑certified gastroenterologists and motility specialists for gastroparesis and gastric‑emptying disorders
- Pharmacologists who can explain GLP‑1 mechanisms, dosing, and causation theories in plain language for judges and juries
- Ophthalmologists and neuro‑ophthalmologists for NAION and other optic‑nerve injuries
- Life‑care planners and vocational experts who quantify future medical needs, work limitations, and long‑term costs
This multidisciplinary team helps us document what happened medically, how it connects to the drug, and what you will likely face in the future, key issues in both settlement negotiations and trial.
Proven Results in Complex Drug Litigation
Our attorneys have collectively recovered millions of dollars for clients in pharmaceutical MDLs and other complex injury cases, including claims against major drug manufacturers. While past results cannot guarantee future outcomes, our experience shows that we:
- Know how to develop cases for bellwether trials and global‑resolution talks
- Understand how to work within MDL structures while preserving your individual story and damages
- Are prepared to stand up to large pharmaceutical companies and their insurers in discovery, motion practice, and negotiation
Our goal in GLP‑1 litigation is straightforward: to position your claim for the strongest possible recovery under the facts and the law.
No Upfront Costs – Ever
We represent GLP‑1 clients on a contingency‑fee basis:
- No attorney fee unless we obtain a recovery for you
- We advance the costs of experts, records, and litigation
- Free, no‑obligation initial consultation
- Clear, written fee agreement from the beginning
You can find out whether you have a potential GLP‑1 or NAION claim without risking a single dollar of your own money.
Comprehensive Case Management
GLP‑1 injury and NAION cases demand detailed documentation. We take on the heavy lifting so you can focus on your health:
- Collecting and organizing medical records, pharmacy data, imaging, and diagnostic test results
- Coordinating with your treating doctors and consulting specialists
- Ensuring you obtain critical diagnostic testing tied to NAION and other alleged GLP‑1 injuries
- Preparing and submitting required MDL forms, plaintiff fact sheets, and authorizations
- Communicating with defendants and insurers and keeping you updated at each major step
- Pursuing maximum compensation for medical bills, lost income, pain and suffering, and future care needs
From intake to resolution, we work to make a complex process as manageable and transparent as possible.
Compassionate, Client‑Centered Support
We understand that sudden vision loss, chronic gastroparesis, and other GLP‑1‑related injuries are life‑changing, not just “cases.” You can expect:
- Direct access to your legal team, not just a call center
- Prompt responses to questions and clear explanations at every stage
- Respect for your circumstances, schedule, and comfort level
- Guidance and support through medical, financial, and legal uncertainty
We also work with MDL Steering Committee counsel and other leadership where appropriate, so your case benefits from the broader discovery record and litigation strategy while still receiving individualized attention.
Contact Us for a Free Consultation
Vision loss may be permanent, but your legal rights are not. Filing deadlines apply.
If you took Ozempic, Wegovy, Mounjaro, or Zepbound and developed sudden vision changes, suspected NAION, or documented optic‑nerve damage, you may have a legal claim against the manufacturers of these GLP‑1 medications. Our team follows the latest medical evidence and court developments in the emerging GLP‑1 NAION vision‑loss MDL and related state‑court proceedings.
Visit www.GLP1Lawsuits.com to:
- Get a free case evaluation
- Speak with an experienced pharmaceutical‑litigation attorney about NAION and GLP‑1 drugs
- Learn more about your legal options in federal MDL proceedings and related state‑court consolidations
- Obtain timeline‑specific analysis of your GLP‑1 use, diagnosis, and vision‑loss history
- Join others seeking justice and accountability for preventable optic‑nerve injuries
Do not wait until the statute of limitations runs out. Courts handling GLP‑1 litigation apply strict filing deadlines that can permanently bar otherwise valid claims if missed. Contact us today to protect your rights and pursue the compensation you deserve. Remember, there are no upfront fees and no costs unless we win your case.
Sources Cited
| # | Authority | Description |
|---|---|---|
| 1 | NIH / StatPearls – NCBI Bookshelf | Kaur K, Margolin E. “Nonarteritic Anterior Ischemic Optic Neuropathy.” StatPearls . Updated September 2025. Comprehensive clinical review of NAION pathophysiology, epidemiology, risk factors, diagnosis, and management |
| 2 | PMC / Eye (London) | Martin-Gutierrez MP, Petzold A, Saihan Z. “NAION or not NAION? A literature review of pathogenesis and differential diagnosis of anterior ischaemic optic neuropathies.” Eye (Lond). 2024;38(3):418–425 |
| 3 | American Optometric Association (AOA) | Clinical report on GLP‑1 receptor agonists and ocular health; public guidance on NAION risk in GLP‑1 users (2025) |
| 4 | World Health Organization (WHO) | Global safety communication: “The use of semaglutide medicines and risk of NAION” (June 2025) |
| 5 | European Medicines Agency (EMA) / PRAC | Pharmacovigilance conclusion: NAION classified as a very rare side effect of semaglutide; label‑update directive (2025) |
| 6 | JAMA Ophthalmology | Matched‑cohort study of 16,827 patients: “Risk of Nonarteritic Anterior Ischemic Optic Neuropathy in Patients Prescribed Semaglutide” (2024) |
| 7 | JAMA Network Open | Target‑trial emulation (~1.5 million patients): “Semaglutide or Tirzepatide and Optic Nerve and Visual Pathway Disorders in Type 2 Diabetes” (2025) |
| 8 | Judicial Panel on Multidistrict Litigation (JPML) – MDL No. 3094 | Transfer orders centralizing GLP‑1 RA Products Liability Litigation; orders on MDL scope and structure (2024) |
Disclaimer
This article provides general information about NAION, GLP‑1 medications, and related litigation and does not constitute medical or legal advice. For diagnosis, treatment, or questions about your medications, consult a qualified healthcare provider such as an ophthalmologist, neuro-ophthalmologist, endocrinologist, or primary‑care physician. For legal guidance, consult a licensed attorney who can evaluate your specific facts, including medical records, prescription history, and applicable deadlines. Each case is unique, and past results do not guarantee future outcomes.
© 2026 GLP1lawsuits.com | All Rights Reserved
Last reviewed: March 2026