The honest answer: for most patients, NAION cannot be fully reversed. But "no cure" does not mean "no hope" - and understanding what the evidence actually says matters both medically and legally.
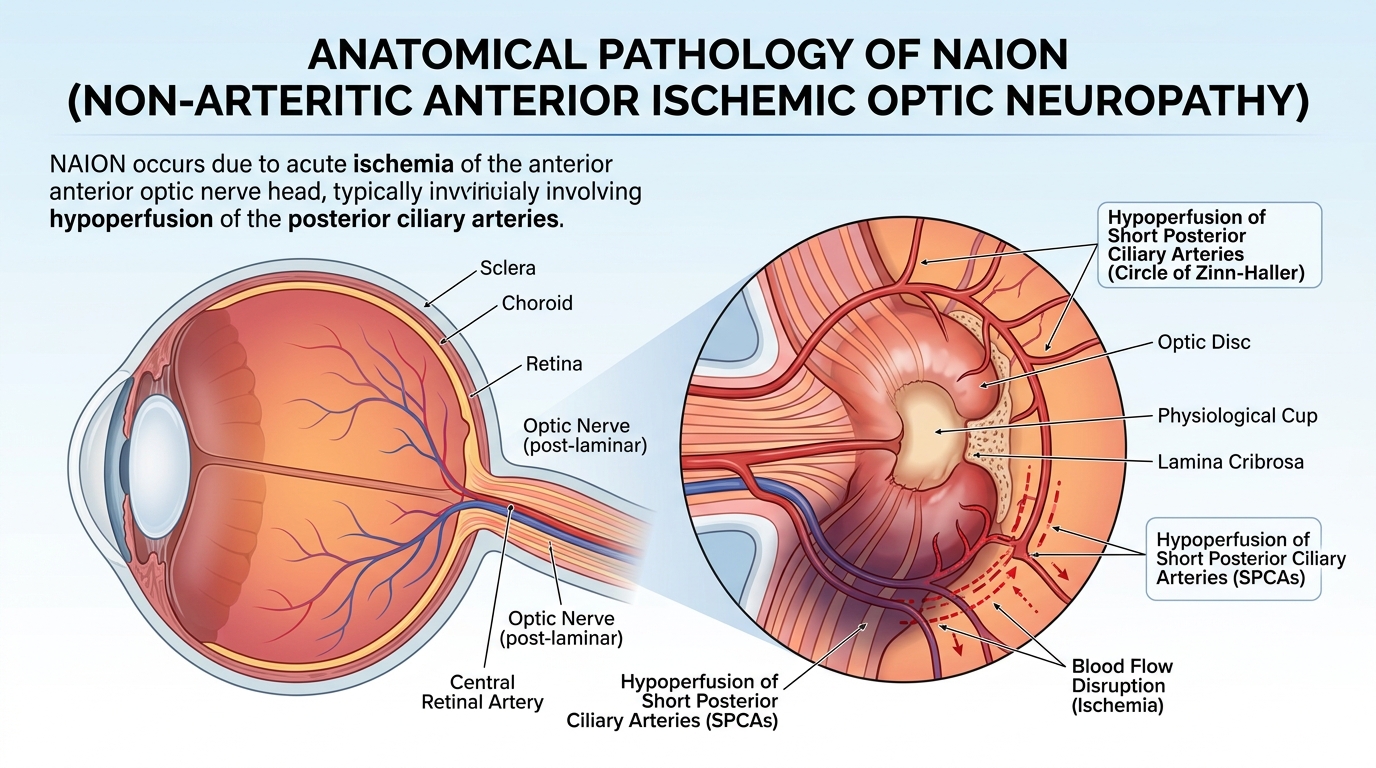
Non-arteritic anterior ischemic optic neuropathy (NAION) is one of the most devastating consequences now being linked to GLP-1 drugs such as Ozempic, Wegovy, Mounjaro, and Zepbound. Once the optic nerve loses blood supply and nerve fibers begin to die, the damage is largely permanent. Yet the full picture of NAION prognosis is more nuanced than a simple “it’s permanent,” and critical developments in 2025 and 2026 suggest the treatment landscape may finally be shifting.
This article covers what the best evidence says about visual recovery, every treatment approach that has been studied, emerging therapies now in pivotal trials, how to protect the unaffected eye, and what prognosis means for your legal claim.
For a full explanation of what NAION is and how it damages the optic nerve, read: What Is NAION? Understanding Non-Arteritic Anterior Ischemic Optic Neuropathy For the link between GLP-1 drugs and NAION, read: Understanding the Growing Concerns Around GLP-1 Drugs and Vision Loss (NAION)
The Hard Truth: No Proven Treatment Reverses NAION
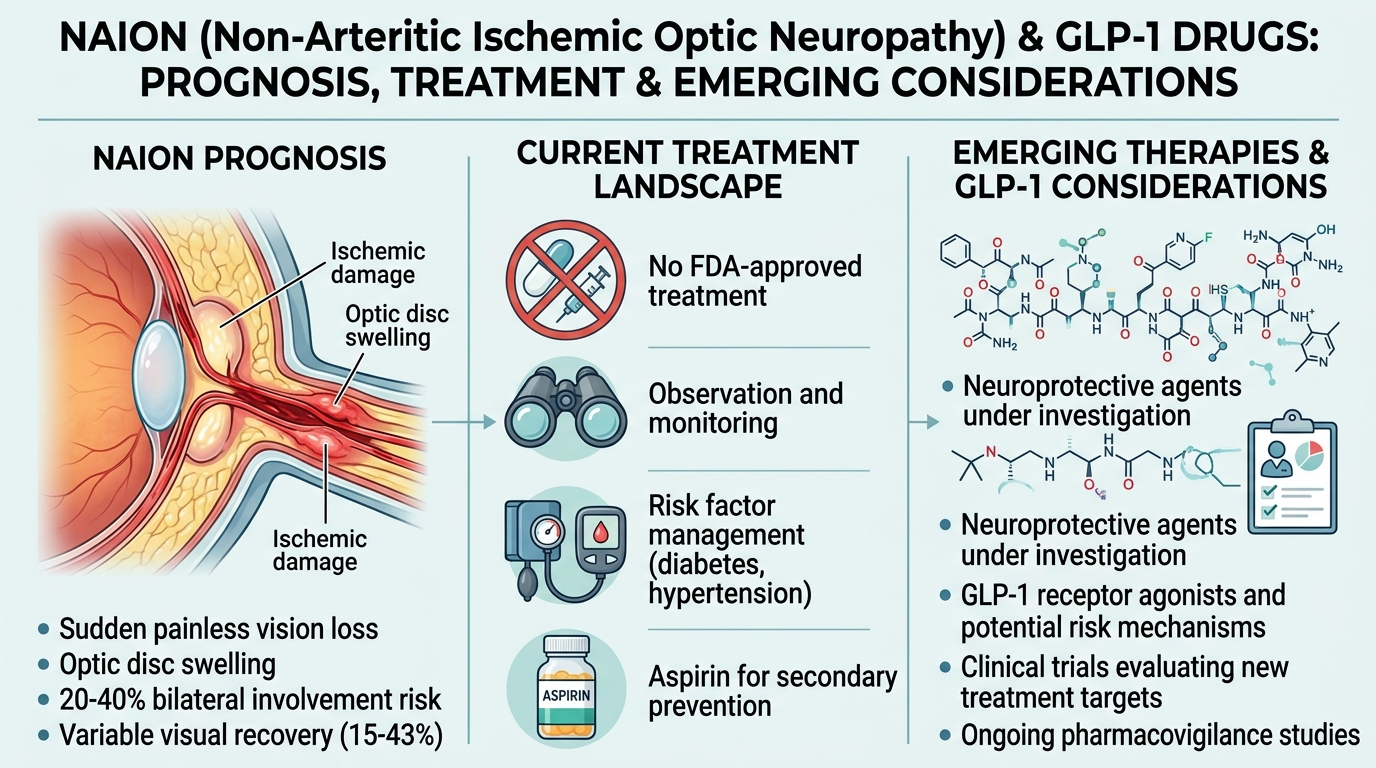
Before walking through the data in detail, patients deserve a clear and honest answer: as of 2026, no medication, surgery, or intervention has been proven in large, well-controlled clinical trials to reliably reverse NAION or restore vision that has been lost.
This is not a failure of effort. Researchers have studied corticosteroids, surgical decompression, anti-VEGF injections, hyperbaric oxygen, levodopa, brimonidine, and other agents over decades. None has demonstrated convincing, reproducible benefit in randomized controlled trials. The current standard of care focuses on stabilizing the condition, protecting the fellow eye, and managing systemic risk factors, not on reversing damage that has already occurred.
This reality is medically significant, legally significant, and deeply personal for patients who experienced sudden vision loss while taking a GLP-1 drug. The permanence of NAION-related injury is central to the damages calculation in ongoing GLP-1 NAION lawsuits.
The Natural History of NAION: What Happens Without Treatment
The IONDT: The Most Important Study Ever Done on NAION Prognosis
The Ischemic Optic Neuropathy Decompression Trial (IONDT) remains the largest and most definitive randomized controlled study of NAION’s natural history. Originally designed to test whether optic nerve sheath decompression surgery could help, the trial’s most important finding turned out to be its control-group data: what happens to NAION patients who receive no active treatment.
The IONDT found, across two years of follow-up:
| Outcome | Percentage of Patients |
|---|---|
| Improved ≥3 lines of visual acuity | ~30% |
| Stable (no significant change) | ~50% |
| Worsened ≥3 lines of visual acuity | ~20% |
This was a landmark finding because before the IONDT, the ophthalmic community largely assumed NAION vision loss was entirely static and non-recoverable. The discovery that roughly 30% of patients spontaneously recover at least three Snellen lines over two years fundamentally changed how the field interprets “treatment” trials, because any new therapy must beat 30% improvement to show true benefit above natural history.
The Hayreh Long-Term Study: A More Detailed Picture
Hayreh’s landmark long-term study of NAION natural history, which followed patients for up to 27 years, added important granularity:
- In patients seen within two weeks of onset with visual acuity of 20/70 or worse, 41% showed improvement by 6 months and this essentially plateaued after 6 months, the window for significant natural recovery is the first 6 months
- In patients with moderate to severe visual field defects, only 26–27% showed field improvement at 6 months to 1 year, with most remaining permanent thereafter
- Approximately 50% of NAION patients present with near-normal visual acuity (20/15 to 20/30) at initial visit, even while having significant visual field loss, underscoring that visual acuity alone is a poor measure of NAION severity
- Worsening beyond the initial event is uncommon but real: approximately 9% of patients with initial acuity ≥20/60 and 18% with initial acuity ≤20/70 showed further deterioration at two years
What This Means in Plain Language
For a patient who experienced NAION while on Ozempic or another GLP-1 drug:
- About 1 in 3 may recover some measurable visual acuity over the first six months, a meaningful minority
- About half will remain stable with neither improvement nor further deterioration
- About 1 in 5 will continue to lose vision despite the acute event having passed
- Visual field defects, which are often more functionally disabling than reduced acuity, are less likely to recover than central visual acuity
- After 6 months, very little additional spontaneous recovery occurs, so the vision status at 6 months post-onset generally represents the patient’s long-term outcome
The phrase “some recovery” also requires context: recovering 3 lines on an acuity chart from 20/200 to 20/100 still leaves a patient with significant, legally relevant permanent visual impairment. Full recovery to pre-NAION vision is uncommon and generally not expected.
Treatments That Have Been Studied, and Why None Is Proven
1. Systemic Corticosteroids (Steroids)
Theory: By reducing optic disc swelling inside the tight scleral canal, steroids might interrupt the compartment-syndrome cascade and prevent further nerve fiber death. The rationale was compelling, and steroids became widely used empirically.
What the evidence shows: A 2019 meta-analysis of 8 studies including 720 eyes concluded that steroids do not significantly improve visual acuity in NAION compared with the natural history. Two randomized controlled trials reached the same conclusion: steroid use did not result in better final visual outcomes. In one large trial, steroid use was associated with potential harms, particularly in diabetic patients (worsened hyperglycemia) and hypertensive patients (elevated blood pressure).
Current status: Most major ophthalmology guidelines do not recommend routine systemic corticosteroid use for NAION. Some neuro-ophthalmologists still use them selectively in acute-phase cases with rapidly deteriorating vision, weighing individual patient risk and benefit, but this is not evidence-based practice.
Special concern for GLP-1 users: Many NAION patients on GLP-1 drugs have diabetes. Administering systemic steroids can cause significant blood sugar spikes and is particularly risky in this population, adding another dimension to the harm caused by a drug-related NAION event.
2. Optic Nerve Sheath Decompression Surgery (ONSD)
Theory: Surgically cutting a window in the optic nerve sheath would relieve pressure, restore blood flow, and prevent further axon loss.
What the evidence shows: The IONDT, the very trial designed to test this procedure, found that optic nerve sheath decompression was no better than careful follow-up and carried a risk of serious complications, including worsened vision in some patients. The surgery is no longer recommended for NAION by any major ophthalmology organization.
Current status: Abandoned as a treatment for NAION.
3. Anti-VEGF Agents (Bevacizumab, Ranibizumab)
Theory: Vascular endothelial growth factor (VEGF) contributes to optic disc edema; blocking it might reduce swelling and limit secondary axon loss.
What the evidence shows: Small case series and pilot studies have investigated intravitreal anti-VEGF injections in NAION. While some individual reports showed modest benefit, no randomized controlled trial has demonstrated reliable visual improvement above the natural history rate. The interventions also carry procedural risks including endophthalmitis.
Current status: Investigational only; not recommended outside of research settings.
4. Levodopa (L-DOPA)
Theory: A small, widely cited early study suggested levodopa (a dopamine precursor used in Parkinson’s disease) might improve vision in NAION, leading to a brief period of enthusiasm.
What the evidence shows: Subsequent studies and reviews have failed to confirm a reliable benefit. The initial study was small and methodologically limited.
Current status: Not recommended as standard treatment.
5. Hyperbaric Oxygen (HBO)
Theory: Delivering high-concentration oxygen in a pressurized chamber could compensate for the ischemic insult and salvage borderline nerve fibers.
What the evidence shows: Only anecdotal reports and very small case series exist. No randomized controlled trial has been conducted. Hyperbaric oxygen involves significant logistical burden, cost, and potential risks.
Current status: Investigational; insufficient evidence to recommend.
6. Brimonidine (Topical Eye Drops)
Theory: This alpha-2 adrenergic agonist eye drop (commonly used for glaucoma) was thought to have neuroprotective properties that might help preserve axons after NAION.
What the evidence shows: Clinical trials have not demonstrated significant benefit in NAION outcomes.
Current status: Not recommended specifically for NAION.
7. Aspirin and Antiplatelet Agents
Theory: Since NAION involves a vascular event, antiplatelet therapy might reduce the risk of a second NAION event in the same or fellow eye.
What the evidence shows: Evidence does not support aspirin as a treatment to improve existing NAION. Some studies suggest aspirin may be modestly associated with a lower risk of fellow-eye NAION, but this has not been definitively established. Many physicians recommend aspirin for systemic cardiovascular risk reduction, a separate, well-supported indication that may have incidental optic-nerve benefit.
Current status: Not proven to reverse or improve NAION; may be considered for systemic cardiovascular risk management in high-risk patients.
A Genuine Breakthrough on the Horizon: Privosegtor (OCS-05)
For the first time in decades, there is real scientific reason for optimism about NAION treatment, and it is directly relevant to GLP-1-affected patients filing claims today.
What Is Privosegtor?
Privosegtor (formerly OCS-05), developed by Oculis, is a neuroprotective small molecule peptoid designed to protect and preserve optic nerve fibers after an acute ischemic or inflammatory event. It works by targeting the ErbB receptor signaling pathway involved in axon survival and regeneration.
Phase 2 Results in Optic Neuritis (ACUITY Trial)
In the Phase 2 ACUITY trial, privosegtor showed statistically significant improvement in visual function and anatomical preservation of optic nerve structures compared to placebo in patients with acute optic neuritis. These results led the FDA to grant privosegtor Breakthrough Therapy Designation in January 2026, the agency’s signal that it may offer substantial improvement over existing therapies.
PIONEER-3: The Pivotal NAION Trial
Oculis has launched the PIONEER program, a series of three pivotal Phase 3 trials supporting registration for optic neuritis and NAION:
- PIONEER-1: Acute optic neuritis in MS patients (began Q4 2025)
- PIONEER-2: Acute optic neuritis without MS (planned H1 2026)
- PIONEER-3: Acute-onset NAION, planned for mid-2026
PIONEER-3 is designed specifically for NAION patients, sharing core design elements with the optic neuritis trials. If successful, privosegtor could become the first approved pharmacological treatment for NAION in history.
What This Means for GLP-1 NAION Patients
This development has two practical implications:
- Medical: Patients experiencing NAION today, including those caused by GLP-1 drugs, may have access to clinical trial enrollment in PIONEER-3, potentially providing the first real treatment option ever available for NAION. Any patient with recent-onset NAION should ask their ophthalmologist about enrollment eligibility.
- Legal: The existence of a developing treatment does not reduce defendants’ liability, if anything, it establishes more clearly that NAION is a serious, recognized medical condition with substantial unmet treatment need, and that the pharmaceutical industry knows the harm is real.
What Can Actually Be Done: The Current Standard of Care
While no proven reversal exists, there is a meaningful and evidence-based management approach that every NAION patient should follow, particularly those whose event may be linked to GLP-1 drugs.
1. Discontinue the Suspected Trigger
The European Medicines Agency (EMA) and World Health Organization (WHO) now formally recommend that semaglutide be discontinued if NAION is confirmed. This is not a guarantee of preventing further deterioration, but it removes the ongoing vascular insult and is the appropriate immediate step. Discuss this with your prescribing physician and your ophthalmologist together.
2. Optimize Blood Pressure; But Carefully
Hypertension is a major risk factor for optic nerve ischemia, so blood pressure control is critical. However, overcorrection, especially at night, can itself trigger or worsen NAION through nocturnal hypotension. Key evidence-based recommendations include:
- Avoid taking antihypertensive medications at bedtime if nocturnal hypotension is suspected
- Consider 24-hour ambulatory blood pressure monitoring to detect dips during sleep
- Target consistent daytime blood pressure control without over-aggressive reduction
3. Treat Obstructive Sleep Apnea Aggressively
OSA is one of the strongest modifiable risk factors for both first and fellow-eye NAION. Non-compliance with CPAP therapy is associated with a 4.5× increased risk of fellow-eye NAION. This is one of the most actionable interventions available:
- Use CPAP consistently every night, compliance, not just diagnosis, matters
- Consider a sleep study if OSA has never been formally evaluated
- Untreated OSA causes repeated nocturnal oxygen drops and blood-pressure swings that directly stress the vulnerable optic nerve
4. Control Diabetes, Cholesterol, and Other Vascular Risk Factors
The optic nerve is a vascular end-organ; its health depends on healthy microvasculature. For GLP-1 users who developed NAION, the same systemic risk factors that may have contributed to the event must now be managed with renewed urgency:
- Optimize HbA1c, but avoid rapid swings in blood sugar
- Control LDL cholesterol with statins if indicated
- Address smoking, obesity, and sedentary lifestyle
- Coordinate care between ophthalmologist, endocrinologist, cardiologist, and sleep specialist
5. Consider Timing of Antihypertensive Medications
A growing body of evidence supports switching antihypertensive medications from an evening dose to a morning dose in NAION patients with hypertension. This aims to preserve normal nocturnal dipping without the exaggerated overnight pressure drops that may compromise optic nerve head perfusion.
6. Monitor the Fellow Eye Closely
Because 15–25% of NAION patients develop the condition in their second eye within five years, protecting the unaffected eye is a medical priority:
- Regular follow-up with an ophthalmologist (at minimum every 6–12 months)
- Prompt evaluation of any new visual symptoms in the fellow eye, do not wait
- Aggressive risk-factor management as above
- Patients with diabetes and severe obstructive sleep apnea face the highest fellow-eye risk
7. Low Vision Rehabilitation
For patients with permanent, significant vision loss, low vision rehabilitation is a critical and underutilized resource. A 2025 PMC study confirmed that structured low vision rehabilitation programs significantly improve:
- Quality of life and visual functioning
- Self-efficacy, patients’ confidence in their ability to manage daily activities
- Independence in tasks like reading, writing, and self-care
- Caregiver burden, formal rehabilitation reduces the strain on family members
Low vision rehabilitation tools and strategies include:
- High-magnification spectacle lenses and stand magnifiers
- Electronic magnification devices and screen readers
- Eccentric viewing training (learning to use remaining peripheral vision)
- Orientation and mobility training for patients with significant field loss
- Driving evaluation and, where vision permits, bioptic telescope systems
- Workplace accommodation planning and vocational rehabilitation
- Psychological support for vision-loss adjustment and depression screening
Low vision rehabilitation is not optional for patients with permanent NAION-related impairment. It should be prescribed by the treating ophthalmologist and quantified by a life-care planner, as its cost and necessity are directly relevant to damage calculations in legal claims.
Prognosis by Patient Profile: Who Recovers More?
Not all NAION prognoses are equal. Evidence suggests the following factors influence recovery potential:
| Factor | Effect on Prognosis |
|---|---|
| Younger age | Better prognosis for visual recovery |
| Initial visual acuity 20/60 or better | More stable course; lower risk of worsening |
| Initial visual acuity 20/70 or worse | More recovery potential (41% improve) but also higher worsening risk (18%) |
| Altitudinal field defect | Usually permanent; less likely to resolve than other field patterns |
| Early presentation (< 2 weeks) | Best window for any intervention and for documenting natural recovery |
| Controlled systemic risk factors | Better long-term stability for fellow eye |
| Continued GLP-1 use post-NAION | Risk of further optic nerve events; EMA/WHO recommend discontinuation |
| Untreated OSA | 4.5× increased risk of fellow-eye involvement |
| Diabetes | Significantly shorter time to fellow-eye involvement (median 14 months vs. 122 months in non-diabetics) |
What NAION Prognosis Means for Your Legal Claim
The prognosis data above is not just medically important; it is foundational to damages in a GLP-1 NAION lawsuit.
Permanent Impairment = Significant Compensable Damages
Because NAION is largely permanent in the majority of patients, and because the vision lost cannot be restored by currently available treatments, the damages in a successfully litigated NAION claim may include:
- Past and future medical expenses: Ophthalmology follow-up, OCT imaging, visual field testing, low vision devices, rehabilitation services, and, if privosegtor or another therapy becomes available, future treatment costs
- Lost wages and earning capacity: Vision impairment that prevents driving, reading, or performing occupational tasks
- Pain and suffering: The psychological and emotional impact of sudden, permanent vision loss, anxiety, depression, and adjustment disorder are well-documented in NAION patients
- Loss of enjoyment of life: Inability to engage in hobbies, recreational activities, and social functions that depend on intact vision
- Life care plan costs: Future medical, therapeutic, and assistive technology needs calculated over the patient’s expected lifetime
Why Prognosis Must Be Documented Thoroughly
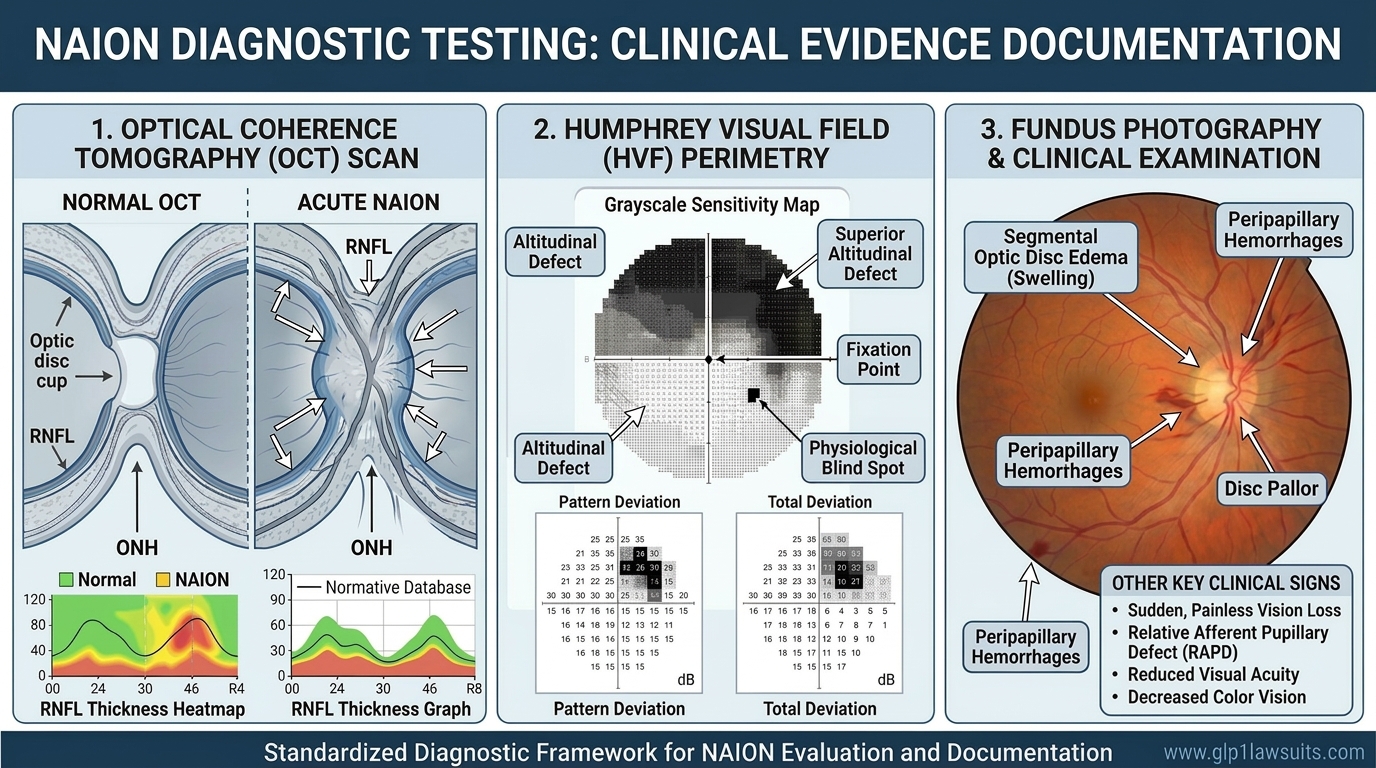
Because some patients experience partial spontaneous recovery, defendants in NAION litigation will argue that vision loss was temporary or incomplete. Thorough, serial documentation, OCT imaging, visual field tests, and functional assessments at multiple time points — is essential to establish the permanent nature and extent of injury.
If you developed NAION while taking a GLP-1 drug, preserving all eye examination records, imaging studies, and functional assessments is critical to building a strong claim.
For detail on what tests document NAION and why they matter legally, read: NAION Diagnosis: What Eye Tests Are Involved? and Diagnostic Tests for NAION and Vision Loss: What to Expect For the latest litigation developments, read: NAION MDL Update
Q&A: NAION Prognosis, Treatment, and Recovery
Q1. Is NAION permanent?
For most patients, yes, the vision lost in a NAION event is largely permanent. Approximately 30% of patients recover at least 3 lines of visual acuity over two years of natural history, but the majority experience stable or worsening vision, and visual field defects (which are often the most functionally disabling aspect) rarely fully resolve. The 6-month mark is generally when most spontaneous recovery has occurred; after that, significant additional improvement is unlikely.
Q2. What is the best treatment for NAION right now?
There is no proven treatment that reliably reverses NAION as of 2026. Current management focuses on: discontinuing suspected triggers (including GLP-1 drugs per EMA/WHO guidance), aggressively treating blood pressure, diabetes, cholesterol, and obstructive sleep apnea, adjusting the timing of antihypertensive medications to prevent nocturnal hypotension, monitoring the fellow eye closely, and low vision rehabilitation for patients with permanent impairment.
Q3. Is there a new treatment coming for NAION?
Yes, the most promising development in decades. Privosegtor (OCS-05) by Oculis received FDA Breakthrough Therapy Designation in January 2026 for optic neuritis, and the pivotal PIONEER-3 trial specifically for NAION is planned for mid-2026. If successful, it would be the first approved pharmacological therapy for NAION in history. Patients with recent-onset NAION should ask their ophthalmologist about clinical trial eligibility.
Q4. Can the other eye also be affected?
Yes. Approximately 15–25% of patients experience NAION in the fellow eye within five years. Patients with diabetes, untreated obstructive sleep apnea, and bilateral disc-at-risk anatomy face the highest risk. CPAP non-compliance in patients with OSA is associated with a 4.5× increased risk of fellow-eye involvement. Aggressive risk-factor management is the primary strategy to protect the remaining eye.
Q5. If some vision returns on its own, does that hurt my legal case?
Not necessarily. Partial spontaneous recovery does not negate the permanent damage that occurred, nor does it eliminate the psychological and functional impact of the event. What matters is thorough documentation of the extent of permanent injury, including OCT-measured nerve fiber layer loss, persistent visual field defects, and functional limitations. A life-care planner can quantify ongoing medical needs regardless of partial recovery. Only a licensed attorney can evaluate how your specific recovery history affects your individual claim.
Q6. Can steroids help my NAION?
The evidence does not support routine steroid use for NAION, multiple meta-analyses and randomized controlled trials have shown no significant benefit on final visual outcomes. Steroids also carry meaningful risks, particularly worsened blood sugar in diabetic patients and elevated blood pressure in hypertensive patients, both very common in GLP-1 drug users. Most major ophthalmology guidelines do not recommend systemic steroids for NAION outside of select clinical judgment cases in the acute phase.
Q7. How do I find a low vision specialist?
Ask your ophthalmologist or optometrist for a referral to a low vision rehabilitation center. The American Academy of Ophthalmology’s provider directory and the American Optometric Association’s low vision resources can also help locate certified low vision specialists. Low vision rehabilitation is covered by many insurance plans including Medicare, and its cost can be included in damages calculations in a GLP-1 NAION legal claim.
Why Work With Us
Exclusive Focus on GLP‑1 Injuries
Our practice is dedicated entirely to GLP‑1 medication injuries, including NAION vision loss, gastroparesis, ileus, and related complications. This is not a side project or add‑on; it is the core of what we do every day.
- Deep, case‑specific knowledge of Ozempic, Wegovy, Mounjaro, Zepbound, and related medications
- Familiarity with how these drugs work, their known side‑effect profiles, and the evolving medical literature around NAION and other serious harms
- Hands‑on experience with MDL procedures, case‑management orders, and plaintiff requirements in complex pharmaceutical litigation
Because we live in this space, we can quickly identify the strengths and weaknesses of a potential GLP‑1 claim and tailor your case strategy around the most up‑to‑date science and MDL developments.
Integrated Medical Expert Network
GLP‑1 and NAION cases are built on medicine as much as law. We work closely with a network of highly qualified specialists who understand these specific injuries:
- Board‑certified gastroenterologists and motility specialists for gastroparesis and gastric‑emptying disorders
- Pharmacologists who can explain GLP‑1 mechanisms, dosing, and causation theories in plain language for judges and juries
- Ophthalmologists and neuro‑ophthalmologists for NAION and other optic‑nerve injuries
- Life‑care planners and vocational experts who quantify future medical needs, work limitations, and long‑term costs
This multidisciplinary team helps us document what happened medically, how it connects to the drug, and what you will likely face in the future, key issues in both settlement negotiations and trial.
Proven Results in Complex Drug Litigation
Our attorneys have collectively recovered millions of dollars for clients in pharmaceutical MDLs and other complex injury cases, including claims against major drug manufacturers. While past results cannot guarantee future outcomes, our experience shows that we:
- Know how to develop cases for bellwether trials and global‑resolution talks
- Understand how to work within MDL structures while preserving your individual story and damages
- Are prepared to stand up to large pharmaceutical companies and their insurers in discovery, motion practice, and negotiation
Our goal in GLP‑1 litigation is straightforward: to position your claim for the strongest possible recovery under the facts and the law.
No Upfront Costs – Ever
We represent GLP‑1 clients on a contingency‑fee basis:
- No attorney fee unless we obtain a recovery for you
- We advance the costs of experts, records, and litigation
- Free, no‑obligation initial consultation
- Clear, written fee agreement from the beginning
You can find out whether you have a potential GLP‑1 or NAION claim without risking a single dollar of your own money.
Comprehensive Case Management
GLP‑1 injury and NAION cases demand detailed documentation. We take on the heavy lifting so you can focus on your health:
- Collecting and organizing medical records, pharmacy data, imaging, and diagnostic test results
- Coordinating with your treating doctors and consulting specialists
- Ensuring you obtain critical diagnostic testing tied to NAION and other alleged GLP‑1 injuries
- Preparing and submitting required MDL forms, plaintiff fact sheets, and authorizations
- Communicating with defendants and insurers and keeping you updated at each major step
- Pursuing maximum compensation for medical bills, lost income, pain and suffering, and future care needs
From intake to resolution, we work to make a complex process as manageable and transparent as possible.
Compassionate, Client‑Centered Support
We understand that sudden vision loss, chronic gastroparesis, and other GLP‑1‑related injuries are life‑changing, not just “cases.” You can expect:
- Direct access to your legal team, not just a call center
- Prompt responses to questions and clear explanations at every stage
- Respect for your circumstances, schedule, and comfort level
- Guidance and support through medical, financial, and legal uncertainty
We also coordinate with MDL Steering Committee counsel and other leadership where appropriate, so your case benefits from the broader discovery record and litigation strategy while still receiving individualized attention.
Contact Us for a Free Consultation
Vision loss is permanent; but your legal rights are not. Filing deadlines apply.
If you took Ozempic, Wegovy, Mounjaro, or Zepbound and developed sudden vision changes, suspected NAION, or documented optic‑nerve damage, you may have a legal claim against the manufacturers of these GLP‑1 medications. Our team follows the latest medical evidence and court developments in the emerging GLP‑1 NAION vision‑loss MDL and related state‑court proceedings.
Visit www.GLP1Lawsuits.com to:
- Get a free case evaluation
- Speak with an experienced pharmaceutical‑litigation attorney about NAION and GLP‑1 drugs
- Learn more about your legal options in federal MDL proceedings and related state‑court consolidations
- Obtain timeline‑specific analysis of your GLP‑1 use, diagnosis, and vision‑loss history
- Join others seeking justice and accountability for preventable optic‑nerve injuries
Do not wait until the statute of limitations runs out. Courts handling GLP‑1 litigation apply strict filing deadlines that can permanently bar otherwise valid claims if missed. Contact us today to protect your rights and pursue the compensation you deserve, no upfront fees and no costs unless we win your case.
Sources Cited
| # | Authority | Description |
|---|---|---|
| 1 | Ischemic Optic Neuropathy Decompression Trial (IONDT) | Landmark NIH-funded randomized controlled trial establishing NAION natural history: ~30% improve, ~50% stable, ~20% worsen at 2 years; decompression surgery found ineffective and potentially harmful (JAMA, Archives of Ophthalmology) |
| 2 | NIH / StatPearls — NCBI Bookshelf | Kaur K, Margolin E. “Nonarteritic Anterior Ischemic Optic Neuropathy.” StatPearls. Updated September 2025 |
| 3 | Hayreh SS — Natural History Study | Long-term NAION natural history: 41% improvement in vision at 6 months for patients ≤20/70; plateau after 6 months; visual field data (Ophthalmology, 2008). PMC2782939 |
| 4 | PMC — Steroids Meta-Analysis | “Steroids in the treatment of nonarteritic anterior ischemic optic neuropathy: a meta-analysis.” 8 studies, 720 eyes; no significant benefit of steroids on visual acuity. PMC6867765 |
| 5 | AAO EyeWiki — NAION | Clinical review: prognosis, management, risk of fellow-eye involvement. Updated November 2024 |
| 6 | IOVS / Karger | Risk factors for fellow-eye involvement in NAION: non-compliance with CPAP (HR 4.5×), diabetes, optic disc drusen; bilateral risk data |
| 7 | Ophthalmology Times / Oculis | FDA Breakthrough Therapy Designation for privosegtor (OCS-05) for optic neuritis; PIONEER program including PIONEER-3 pivotal trial for NAION (January 2026) |
| 8 | Pharmaphorum | Oculis takes privosegtor into pivotal trials; PIONEER-3 NAION trial planned mid-2026 (March 2026) |
| 9 | PMC — Low Vision Rehabilitation | “The effect of low vision rehabilitation on the quality of life and self-efficacy of low vision patients.” PMC11734459 (2025) |
| 10 | American Optometric Association (AOA) | Clinical guidance on GLP-1 receptor agonists and ocular health; NAION risk and monitoring recommendations (2025) |
| 11 | World Health Organization (WHO) / EMA | Safety communications classifying NAION as a very rare side effect of semaglutide; recommendation to discontinue semaglutide if NAION confirmed (June 2025) |
| 12 | Judicial Panel on Multidistrict Litigation (JPML): MDL No. 3163 | New MDL for GLP-1 NAION vision-loss claims (December 2025); separate from GI MDL No. 3094 |
Disclaimer
This article provides general information about NAION prognosis, treatment options, and related GLP-1 litigation and does not constitute medical or legal advice. For diagnosis, treatment decisions, or questions about your specific medications and vision, consult a qualified healthcare provider such as an ophthalmologist, neuro-ophthalmologist, or primary-care physician. For legal guidance, consult a licensed attorney who can evaluate your specific facts, including medical records, prescription history, and applicable filing deadlines. Each case is unique, and past results do not guarantee future outcomes.
© 2026 GLP1lawsuits.com | All Rights Reserved
Last reviewed by a licensed attorney: March 2026